What is Ocaliva - Obeticholic Acid and what is it used for?
Ocaliva is used to treat adult patients with a liver disease known as primary biliary cholangitis, an autoimmune condition in which there is a gradual destruction of the small bile ducts in the liver. These ducts carry a liquid called bile from the liver to the intestine, where it is used to aid in the digestion of fats. As a result of damage to the ducts, bile builds up in the liver causing liver tissue injury. This can lead to scarring and liver failure. , and may increase the risk of liver cancer.
Ocaliva contains the active substance obeticholic acid. This is used in combination with another medicine, ursodeoxycholic acid (UDCA), in patients who do not respond sufficiently to UDCA alone, and on its own in patients who cannot take UDCA.
Because the number of patients with primary biliary cholangitis is low, the disease is considered 'rare', and Ocaliva was designated an 'orphan medicine' (a medicine used in rare diseases) on 27 July 2010.
How is Ocaliva used?
Ocaliva is available as tablets (5 and 10 mg) and can only be obtained with a prescription. The recommended starting dose is 5 mg once a day. If Ocaliva is well tolerated after six months, the dose can be increased to 10 mg once daily. Doses may be decreased or treatment may need to be stopped in patients who experience intolerable itching (a possible side effect of Ocaliva).
For more information, see the package leaflet.
How does Ocaliva - Obeticholic Acid work?
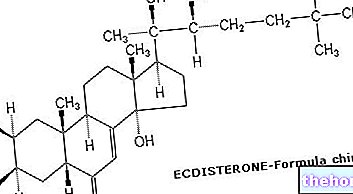
The active substance in Ocaliva, obeticholic acid, is a modified form of a bile acid (bile acids are the main components of bile). It works mainly by attaching to receptors in the liver and intestines (farnesoid X receptors) that control the production of bile. By attaching to these receptors, Ocaliva reduces the production of bile in the liver, preventing it from accumulating and damaging liver tissue.
What benefit has Ocaliva - Obeticholic Acid shown during the studies?
Ocaliva was compared with placebo (a dummy treatment) in one main study involving 217 adults with primary biliary cholangitis who had been taking UDCA for at least 1 year or who could not take UDCA. The measure of effectiveness was based on the number of patients whose blood levels of substances such as bilirubin and ALP (markers of liver damage) had decreased by at least 15% (for ALP) and below a certain value considered normal (for bilirubin) after 1 year of treatment.
The study showed that Ocaliva was more effective than placebo in reducing blood levels of bilirubin and ALP: levels decreased by the amount required in 47% (34 out of 73) of patients treated with Ocaliva 10 mg and in 46% (32 out of 73) 70) of patients treated with increasing doses of Ocaliva (5 mg to 10 mg), compared with 10% (7 of 73) of patients treated with placebo.
What are the risks associated with Ocaliva - Obeticholic Acid?
The most common side effects with Ocaliva are itching (which may affect more than 6 in 10 people) and tiredness (which may affect more than 2 in 10 people). Itching is also the most common side effect that can lead to discontinuation of treatment. In most cases observed, itching occurred within the first month of treatment, tending to resolve over time while continuing treatment. "Full list of side effects reported with Ocaliva, see package leaflet.
Ocaliva must not be used in patients with completely blocked bile ducts. For the full list of restrictions, see the package leaflet.
Why has Ocaliva - Obeticholic Acid been approved?
Patients with primary biliary cholangitis have limited treatment options. Ocaliva has been shown to reduce blood levels of bilirubin and ALP in patients with primary biliary cholangitis, including those who could not be treated with UDCA. The reductions in bilirubin and ALP were found to be such as to be indicative of an improvement in the condition of the liver. However, the clinical benefits of Ocaliva need to be demonstrated in further studies. The safety profile of the medicine was considered to be favorable, with side effects that were tolerable and manageable with supportive treatment (e.g. for pruritus) and dose adjustments. The Agency's Committee for Medicinal Products for Human Use (CHMP) therefore decided that Ocaliva's benefits are greater than its risks and recommended that it be approved for use in the EU.
Ocaliva has been given "conditional approval". This means that more information on the medicine will be available in the future, which the company is required to provide. Each year the European Medicines Agency will review the new information available and this summary will be updated accordingly.
What information is still awaited for Ocaliva?
Since a conditional approval has been given for Ocaliva, the company that markets Ocaliva will provide additional data from two studies to confirm the medicine's efficacy and safety. The first study is designed to demonstrate the clinical benefit of Ocaliva, while the second study will evaluate the benefits of Ocaliva in patients with moderate to severe liver disease.
What measures are being taken to ensure the safe and effective use of Ocaliva - Obeticholic Acid?
The recommendations and precautions to be observed by healthcare professionals and patients for Ocaliva to be used safely and effectively have been set out in the summary of product characteristics and package leaflet.
More information about Ocaliva - Obeticholic Acid
For the complete version of Ocaliva's EPAR, consult the Agency's website: ema.europa.eu/Find medicine / Human medicines / European public assessment reports. For more information about Ocaliva therapy, read the package leaflet (included with the EPAR) or contact your doctor or pharmacist.
The summary of the opinion of the Committee for Orphan Medicinal Products for Ocaliva is available on the Agency's website: ema.europa.eu/Find medicine / Human medicines / Rare disease designation.
The information on Ocaliva - Obeticholic Acid published on this page may be out of date or incomplete. For a correct use of this information, see the Disclaimer and useful information page.