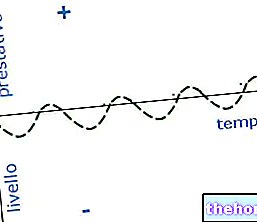
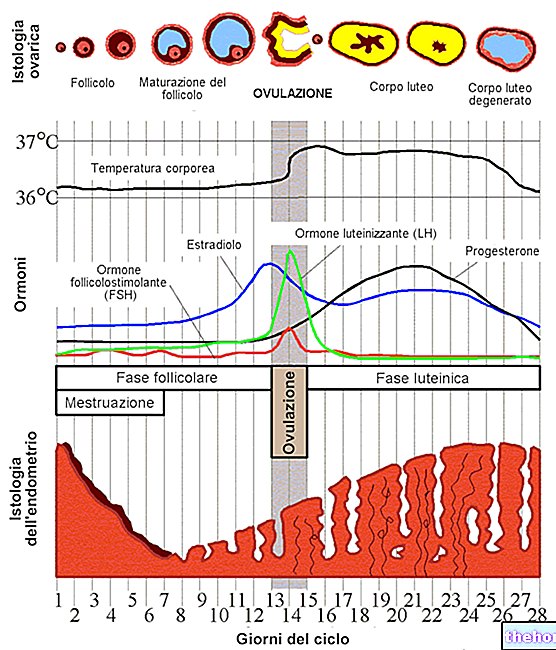
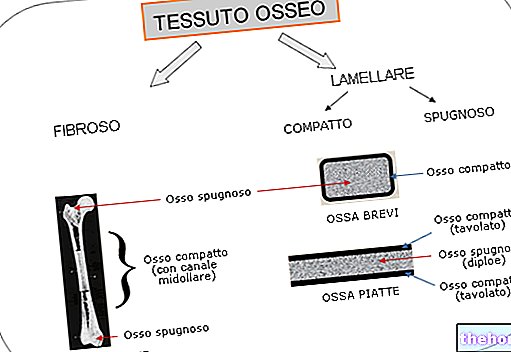
LIPOPHILIA: property possessed by some substances to blend well in fats, oils, lipids and non-polar solvents such as hexane or toluene.

Often the term lipophilic is used as a synonym for hydrophobic or apolar (not soluble in water). In reality there are rare cases of substances, such as silicones and fluorocarbons, which are hydrophobic but not lipophilic.