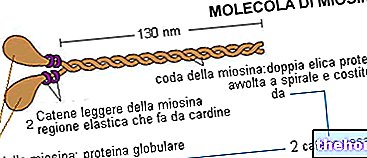
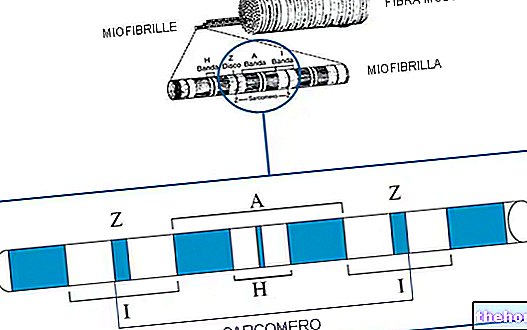
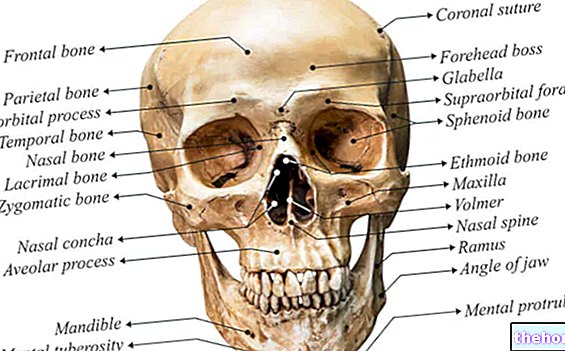
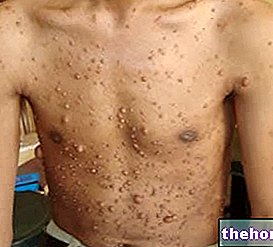
" first part
The enzyme that catalyzes the fourth stage of the krebs cycle is the α-keto glutarate dehydrogenase; this enzyme is an enzyme complex very similar to pyruvate dehydrogenase. Both are composed of 48-60 proteins in which three different enzymatic activities are recognized and also have the same enzymatic cofactors; are very similar enzymes because they act on similar substrates: both pyruvate and l "α- keto glutarate, are α- keto acids. The mechanism of action of the two enzymatic complexes is the same.
The attack by thiamine pyrophosphate on the carbonyl (C = O) of the "α-ketoglutarate, leads to its decarboxylation and the carboxyhydroxy propyl derivative is formed. With the subsequent transfer to the lipoamide, an internal redox process takes place, from which the lipoamide carboxy-derivative or succinyl lipoamide is obtained.
The succinyl lipoamide then reacts with coenzyme A to give succinyl coenzyme A (which continues in the krebs cycle) and the reduced lipoamide which is reoxidized by the FAD: the FADH2 that is formed is reoxidized by NAD + and NADH is obtained . In this stage, therefore, the second elimination of a carbon from the carbonaceous skeleton took place, in the form of carbon dioxide.
An acyl group linked to coenzyme A is in an activated form, that is, it has a high energy content: it is therefore possible to exploit the energy of succinyl coenzyme A.

In the fifth stage of the krebs cycle, succinyl coenzyme A is subjected to the action of succinyl thiokinase; two hypotheses have been made about its mode of action: we will describe only one of the two because it is the most accredited.According to this hypothesis, succinyl coenzyme A is attacked by the nitrogen of a histidine (Hys) of the enzyme: coenzyme A is released and an adduct derived from histidine is formed as an intermediate, that is the succinyl-enzyme (or succinyl-Hys ); an orthophosphate acts on this intermediate, leading to the release of succinate and the formation of the phosphoenzyme. The phosphoenzyme, attacked by guanosine diphosphate (GDP), produces guasnosine triphosphate (GTP) and the enzyme is released. From the energy point of view GTP = ATP: the bond that provides energy is the same in both species (it is the anhydride bond between phosphoryl Β and phosphoryl γ). In some cases GTP is used as a material with a high energy content but, usually, GTP is converted into ATP by the action of the enzyme nucleoside diphospho kinase (NDPK); is an enzyme found in cells and catalyzes the following reaction:
N1TP + N2DP → N1DP + N2TP
Generic NiTP ® nucleoside triphosphate
Generic NiDP ® nucleoside diphosphate
It is a reversible reaction; in our case it happens:
GTP + ADP → GDP + ATP
therefore it can proceed to the right or to the left even for small variations in the concentrations of the reagents.
If the krebs cycle proceeds at such a speed as to lead to a production of ATP higher than the energy requirement, there is a scarce availability of ADP while there is a lot of ATP: the reaction catalyzed by the nucleoside diphospho kinase is, then, directed to the left ( GTP accumulates if the nucleoside diphospho kinase does not have sufficient substrate i.e. ADP). GTP is therefore a signal of energy availability and therefore slows down the krebs cycle.
The sixth stage of the krebs cycle leads to the formation of the fumarate by the action of succinate dehydrogenase; this enzyme gives a stereospecific reaction as the unsaturated (it is an alkene) trans is always formed, ie the fumarate (while the cis isomer is the maleate). Succinate dehydrogenase is found on the inner mitochondrial membrane, while all the other enzymes of the krebs cycle are scattered throughout the mitochondrion.
Succinate dehydrogenase has FAD as a cofactor; it is inhibited by oxaloacetate (feed-back inhibition) while it has succinate and fumarate as a positive modulator (activator). its activator. Let's try to understand why, by jumping to the final stage of the krebs cycle. The final stage of the krebs cycle requires energy so the only possibility to obtain oxaloacetate from the patient is that the concentration of the patient is very high : malate is one of the metabolites with the highest concentration in cells. The reaction that converts malate into oxaloacetate is also favored by the fact that the concentration of oxaloacetate is kept low by the action of citrate synthase. The reaction catalyzed by succinate dehydrogenase is, then, a self-feeding reaction and this is the only way to make the transformation of malate into oxaloacetate take place.
The concentration of mitochondrial malate must be compatible with the concentration of the cytoplasmic malate: only when the concentration of mitochondrial malate is so high as to guarantee the conversion of malate into oxaloacetate (in the krebs cycle) then the malate can also be used in other ways (which are cytoplasmic): in the cytoplasm the malate can be converted into oxaloacetate from which aspartate can be obtained by the action of GOT (it is a transaminase) or glucose through gluconeogenesis.
We return to the seventh stage of the krebs cycle it is catalyzed by the enzyme fumarasi: water is added in a stereospecific way to make L-malate.
In the last stage of the Krebs cycle, of which we have already spoken, the action of the malate dehydrogenase. This enzyme uses a NAD + molecule for its catalytic action.
We have thus concluded the description of the various stages of the krebs cycle.
The krebs cycle is totally reversible.
To increase the speed of the krebs cycle, the concentration of metabolites present in that cycle can be increased; one of the strategies to increase the speed of the krebs cycle consists in converting part of the pyruvate that enters the mitochondria into oxaloacetate (by the action of pyruvate carboxylase) and not transforming it all into acetyl coenzyme A: thus increases the concentration of oxaloacetate which is a metabolite of the krebs cycle and, therefore, increases the speed of the whole cycle.
In the krebs cycle three NAD + are converted into three NADH and one FAD into FADH2 and, moreover, a GTP is obtained: by channeling the reducing power obtained from the krebs cycle, further ATP is produced; in the respiratory chain, the reducing power is transferred from NADH and FADH2 to oxygen: this transfer is due to a series of enzymes located on the mitochondrial membrane which, in their action, lead to the production of ATP.
The processes of the respiratory chain are exergonic processes and the liberated energy is used to produce ATP; the purpose of the cell is to exploit the exergonic processes to make the synthesis of ATP take place. For each molecule of NADH that enters the respiratory chain, 2.5 molecules of ATP are obtained and for each FADH2 1.5 molecules of ATP are obtained; this diversity is due to the fact that FADH2 enters the respiratory chain at a lower level than NADH.
With the reducing power of aerobic metabolism, 30-32 ATP (219-233 kcal / mol) are obtained with an efficiency of about 33% (the efficiency of anaerobic metabolism is about 2%).










.jpg)

.jpg)




-funzione-e-uso-clinico.jpg)