Water and a diet rich in vegetables are very useful for preventing dehydration, but when this has already arisen or there is a real risk that it will appear (severe sweating, etc.) it is much better to sip specially formulated drinks. In fact, water alone could not only be insufficient but even contraindicated (a possible cause of hyponatremia).
The osmolarity of plasma, which under normal conditions is between 280 and 330 mOsm / kg, is mainly influenced by some of its components, such as sodium, proteins and glucose.
To be defined as isotonic, a drink must have the same osmolarity as plasma (while changing the type of solutes). On the contrary, hypotonic and hypertonic are defined as those drinks having, respectively, a lower or higher osmolarity.
The consumption of beverages hypotonic, like water and in particular low residue water, decreases plasma osmolarity (dilutes blood solutes) and tends to reduce the sensation of thirst before water levels are restored.
Beverages hypertonic, on the other hand, due to the laws of osmosis, they draw liquids into the intestinal lumen, aggravating dehydration and becoming a source of possible intestinal disorders (diarrhea).
Having established that the ideal drink to combat dehydration must be isotonic or slightly hypotonic, it is good to remember that most sports drinks comply with this directive (in this case the "isotonic" or "isosmolar" adjective is clearly indicated on the label) .
A healthy and tasty rehydrating drink can also be prepared by mixing 5 tablespoons of table sugar for each liter of water and adding a pinch of salt (1g) and 100 ml of concentrated orange juice.
Furthermore, on the shelves of supermarkets or in specialized shops, there are powdered saline supplements that must always be added to drinks in the dosages indicated on the label. When these are exceeded, in fact, the high salt concentration draws liquids in the intestine by osmotic gradient, removing them from the plasma, causing diarrhea and aggravating the state of body dehydration.
Sweat is an aqueous solution that contains minerals, such as sodium, chlorine, magnesium and potassium.
Sweat is a hyposmotic liquid, that is, with an osmolarity (concentration of the particles present in solution, or solutes) lower than that of all other body fluids; sweat, in fact, has a concentration of 80-180 mOsm / l compared to 280-330 mOsm / l of plasma.
Sweating, therefore, always leads to a loss of water in excess of the loss of salts.
For what has been said, it is good that the rehydrating drink has a concentration equal (isotonic) or slightly lower (hypotonic) to that of the plasma.

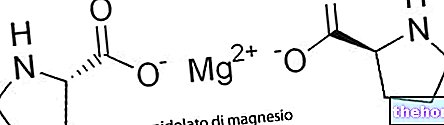








.jpg)