From the same plant it is possible to obtain different essential oils from different organs; for example, we have seen how from bitter orange essential oils are obtained from fruits by pressing, but also from flowers and leaves by steam distillation.
Essential oils are the product of extraction of compounds enclosed in the internal secretion structures (lysigenic, schizolysis, schizolysogen pockets and channels) and external (hairs, scales and emergencies) of the plant. Essential oils are obtained from fresh drugs, which, depending on the organ in which the oil is contained, must be properly prepared, left whole or crushed if they are particularly leathery, or where essential oils are enclosed in internal secretion organs ( conifers and camphor). The extracted substances are mainly monoterpenes and sesquiterpenes; essential oils contain in minimal quantities (1-2%) also flavonoids, and coumarins, compounds also characterized by a low molecular weight. Terpenes can be classified according to their chemical characterization in:
Hydrocarbons consisting of carbon and hydrogen (-CH-);
Hydrocarbons with different levels of oxygenation (-CHO-).
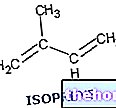
In general, terpenes are molecules containing multiples of 5 carbon atoms. Monoterpenes, for example, are molecules at 10C, with different cyclic characterizations, spatial and optical configurations.
The hydrocarbon monoterpenes and sesquiterpenes (15C) are the main components of essential oils, which in this case have marked irritating properties for the mucous membranes, because not being oxygenated they have greater affinity with the lipid components of the membranes and skin; examples are limonene from citrus fruits and linalool from camphor.
Diterpenes are rarely extracted by steam distillation, since they have a high molecular weight; they are extracted only when the extraction temperatures are very high.
Of the mono and sesquiterpene compounds there are also oxygenated variants, among these we find: alcohols (linalool and genariol), aldehydes (cinnamic and citral aldehyde), ketones (menton), phenols (thymol and eugenol), esters, peroxides [they are very reactive and for their oxidative capacity confer antiseptic or antimicrobial properties to essential oils (escaridiol)], organic acids (cinnamic and benzoic acids) and others; in any case, they are 10C (mono) and 15C (sesqui) molecules with different functionalities. There are also mono and sesquiterpenes with sulfur portions, typical of the Liliaceae.
Essential oils are also classified on the basis of the chemical component that characterizes them on a functional level: essential oils Ad aldehydes, o.e. A phenols, o.e. A ketones, o.e. A peroxides.
All essential oils, regardless of chemical classification, can be processed or treated to change their chemical characteristics and make them purified or activated essential oils.
Purified essential oils: they are deprived of the hydrocarbon component and subjected to treatments aimed at eliminating or significantly reducing the mono and sesquiterpenic component, to reduce the irritating capacity. Purification is carried out by fractional distillation, an extraction process that allows us to selectively extract a certain chemical category from the mixture, which responds - in the transition from the liquid state to the gaseous state - to precise conditions of temperature and pressure: in this case those mono and sesquiterpene with hydrocarbon characterization. The purified essential oil acquires the functional characteristics that allow it to be inserted into a formulation.
Activated essential oils: these are oils in which the oxygenated component is increased with the ultimate aim of enhancing their antimicrobial, antiseptic and disinfectant value. In particular, activated essential oils are rich in peroxides. Logically, they must be included in the formulation of products whose main expression is antiseptic or antimicrobial activity. Essential oils are enriched in peroxides through a process based on the insufflation of air rich in ozone (O3); oxygen, being in itself a particularly reactive element, thus creates functional peroxidation elements, especially on those mono- and sesquiterpene hydrocarbon compounds.
Other articles on "Plants rich in essential oils"
- Drugs to essential oils
- Pharmacognosy
- Essential oils