Always remaining in the biogenetic path of mevalonic acid, let's consider the TERPENIC COMPOUNDS.

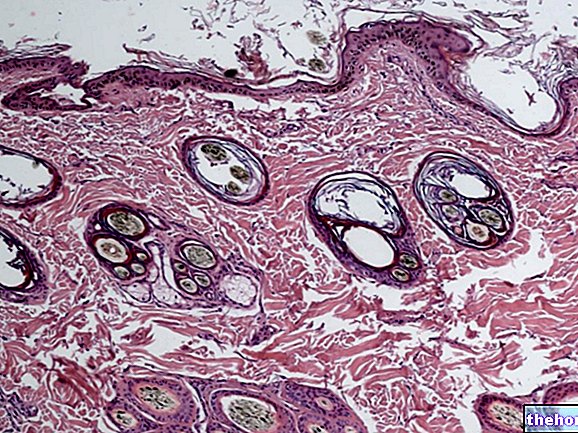
Terpene compounds are ubiquitous: those with lower molecular weight, mono and sesquiterpenes, we find them, for example, in secretory tissues, in which there are mainly molecules of a terpenic nature that can coexist with molecules of another nature, or even with molecules higher molecular weight terpene. Conifer resins are so called because their consistency is semi-solid and they have an aromatic odor; this consistency becomes solid if the resins remain in the air, due to various factors, one of them being the evaporation of the molecules with the lowest molecular weight, therefore volatile: the monoterpenes; the high molecular weight molecules, on the other hand, remain and are represented by terpenes, such as triterpenes or tetra terpenes.
In secreted tissues there are mainly terpenic compounds, which can be low or high molecular weight; this distinguishes the resins of a semi-solid nature from the essential oils contained in the glandular hairs of a plant.
An essential oil is liquid because it does not contain high molecular weight terpenes; a resin is solid because, in addition to containing mono and sesquiterpenes, it also contains higher molecular weight terpenes.
Secretion tissues are not the only ones to contain terpene compounds; for example, an essential oil is not obtained from the Gentian, but the plant still contains essential oils. Furthermore, essential oils are not the only naturally occurring terpene compounds, and terpenes can characterize drugs that do not have an essential oil at all; we remember in fact that terpenes are ubiquitous compounds.
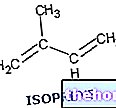
Terpenes derive from isoprene (C5), which is phosphorylated to ensure it enters a biochemical pathway of synthesis, that of terpenes. The phosphorylation of isoprene implies an energetic activation of the molecule (the phosphoric bond is a high energy bond); when isoprene is phosphorylated it can assume different optical conformations, but we are interested in the molecule maintaining a 5-carbon structure, because that is the building block that allows all the terpene-type molecules to be built. The phosphorylated isoprene can be called in two different ways: isopentenyl-pyrophosphate or dimethylallyl-pyrophosphate; the molecule is the same, but with different optical conformations. When a molecule of isopentenyl-pyrophosphate joins a molecule of dimethylallyl-pyrophosphate, a molecule of geranyl-pyrophosphate is produced; a pyrophosphate group (2 phosphors) detaches from one of the two isoprene units, releasing the energy that united the two C5 units.
Geranyl pyrophosphate is a molecule with 10 carbon atoms; it is the precursor of monoterpenic compounds because it has in itself the architecture and the number of carbons suitable for that chemical category. The geranyl-pyrophosphate then in turn loses the pyrophosphate group and becomes the starting point to give rise to any of the thousands of known monoterpenes: camphor, menthol, eucalyptol, pinene. These molecules, regardless of their cyclic or linear configuration, as well as the substituents they will present, will always be C10 molecules, for this reason attributable to the family of monoterpenes. Geranyl-pyrophosphate however, it can in turn be a precursor of successive terpene categories, and if it remains phosphorylated it can bind another molecule of isopentenyl-pyrophosphate or dimethylallyl-pyrophosphate and thus become the precursor of sesquiterpene compounds. The union of geranyl-pyrophosphate with another unit at C5 releases a pyrophosphate, giving the energy necessary for a new condensation, with the obtainment of a final product called farnesyl-pyrophosphate. This last is a molecule with 15 atoms of carbon which is a precursor of sesquiterpenes, compounds which - beyond their aliphatic or cyclic configuration and the degree of substitution - have 15 carbon atoms. Farnesyl-pyrophosphate can in turn be a precursor of sesquiterpene compounds with a higher molecular weight; has 15 carbon atoms and these molecules can undergo condensation two by two, thus forming molecules of C30 (precursors of triterpene or sterol compounds, where a sterol is a C27 because it is decarboxylated). However, farnesyl-pyrophosphate can once again bind to another C5 unit, forming geranyl-geranyl-pyrophosphate, precursor of molecules with 20 carbon atoms, called diterpenes. From the further condensation of two C20 units, therefore of two molecules of geranyl-geranyl-pyrophosphate, tetra terpenes are obtained, molecules consisting of C40. Tetra terpenes give extremely important molecules, such as carotenoids and vitamin E, pigments with important antioxidant properties, highly sought after on the wellness market, increasingly oriented towards the search for the natural at any cost.
Other articles on "Terpenes"
- Butcher's broom and Centella
- Pharmacognosy
- Drugs to essential oils