Active ingredients: Vitamins
Cernevit Powder For Injectable Solution
Why is Cernevit used? What is it for?
Pharmacotherapeutic group:
Polyvitamin for parenteral administration
Therapeutic indications:
Vitamin intake corresponding to the daily requirement of the adult and child over 11 years of age, in situations requiring an injectable vitamin supplement when the "oral intake" is contraindicated, impossible or insufficient (malnutrition, digestive malabsorption , parenteral nutrition ......).
Contraindications When Cernevit should not be used
Hypersensitivity "known to one of the components of this specialty"; in particular, do not inject people with a history of intolerance to thiamine (vitamin B1).
Pregnancy.
Precautions for use What you need to know before taking Cernevit
Given the presence of vitamin A (retinol) in this specialty, "take into account the doses administered in case of combinations with other preparations already" containing this vitamin. CERNEVIT does not contain vitamin K. The "latter will" need to be given separately if needed.
After IV bolus injection, a moderate elevation of SGPT transaminases only was observed in some patients with developmental inflammatory enterocolitis; rapidly reversible increase on discontinuation of dosing. It is therefore recommended that transaminase levels be monitored in this type of patient. Due to the presence of glycolic acid, repeated and prolonged administration in patients with jaundice of hepatic origin or important biological cholestasis requires careful monitoring of liver functions.
Interactions Which drugs or foods can modify the effect of Cernevit
vitamin B6 can antagonize the therapeutic effect of levo dopa.
Warnings It is important to know that:
Very high doses of vitamin A in animals are teratogenic and have been associated in isolated cases with malformations in humans. Daily amounts of vitamin A higher than 10,000 IU should be avoided during pregnancy (especially in the first months) and a doctor should be consulted. for advice on the total amount of vitamin A that can be taken through the different sources.
The product can be taken without risk by people with celiac disease.
Dosage and method of use How to use Cernevit: Dosage
Adults and children over 11 years of "age":
1 bottle per day
Particular posologies: When an increased supply of nutrients is required (for example severe burns) CERNEVIT can be administered at daily doses 2-3 times higher than those normally used.
Mode "d" use
Intravenously
CERNEVIT can "enter the composition of nutritional mixtures in association with carbohydrates, lipids, amino acids, electrolytes, after verifying the compatibility" and established "of each type of mixture.
Intramuscularly
Dissolve the lyophilisate in 2.5 ml of solvent (water for injections), as mentioned above.
Cernevit (bottle without BIO-SET)
With a syringe, inject the contents of a solvent vial (5 ml of water for injections) into the bottle. Shake lightly to dissolve the lyophilisate.
E.V. slow or perfusion in saline, glucose, etc.
Cernevit BIO-SET
Cernevit BIO-SET allows reconstitution directly in bags equipped with an injection site (whether they are single or multi-compartment bags). See Figure 1
Single bag:
- Remove the cap by twisting and pulling it to break the safety ring;
- Connect the BIO-SET directly to the bag through the injection site;
- Activate the BIO-SET by applying light pressure on the transparent mobile part of BIO-SET. This action pierces the rubber stopper of the bottle.
- Place the combined system (Cernevit BIO-SET + the infusion bag) vertically with the bag on top. Squeeze the bag lightly several times to transfer the solution into the bottle (approximately 5 ml). Shake the bottle to reconstitute Cernevit.
- Turn the combined system upside down by bringing the bag down. Lightly squeeze the infusion bag several times to push air into the bottle headspace allowing the solution to be transferred into the bag.
- Repeat steps 4 and 5 until the bottle is empty.
- Remove and discard the Cernevit BIO-SET.
Multi-compartment bag:
Reconstitution of Cernevit BIO-SET must be done before activating the multi-compartment bag (before opening the non-permanent membranes and before mixing the contents of each compartment).
- Place the multi-compartment bag on a work surface;
- Remove the cap of Cernevit BIO-SET by turning and pulling it to break the safety ring;
- Connect the BIO-SET directly to the multi-compartment bag through the injection site;
- Activate the BIO-SET by applying light pressure on the transparent mobile part of BIO-SET. This action pierces the rubber stopper of the bottle;
- Hold the bottle vertically. Squeeze the bag lightly several times to transfer the solution into the bottle (approximately 5 ml). Shake the bottle to reconstitute Cernevit.
- Turn the combination system upside down while holding the bottle upside down. Lightly squeeze the infusion bag several times to push the air into the bottle headspace allowing the solution to be transferred into the bag.
- Repeat steps 5 and 6 until the bottle is empty.
- Remove and discard the Cernevit BIO-SET.
- Finally, activate the multi-compartment bag.
Attention:
Make sure that the BIO-SET is always connected to the injection point throughout the reconstitution process.
Overdose What to do if you have taken too much Cernevit
Manifestations of hypervitaminosis A and hypervitaminosis D (symptoms linked to hypercalcemia) are possible in case of prolonged administration of large doses of these vitamins.
Side Effects What are the side effects of Cernevit
Sporadic rise in SGPT transaminases after IV injection in some subjects (see: "Precautions for use"). Given the presence of vitamin B1, it is possible to observe anaphylactic reactions in subjects with latent allergy (see: Contraindications). Possibility of pain at the site of intramuscular injection, in which case a slow intravenous or deep intramuscular injection is recommended.
The patient is invited to communicate any undesirable effect other than those indicated above to his doctor or pharmacist.
Expiry and Retention
Store at a temperature below 25 ºC;
Store in the original package to protect the medicine from light
Expiration:
Do not use the medicine after the expiry date indicated on the package.
Keep out of the reach of children.
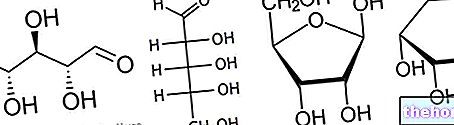
Composition
Each bottle of Powder for Solution for Injection contains:
Pharmaceutical form
Powder For Injectable Solution
Packs
1 bottle of Powder For Injectable Solution
10 vials of Powder For Injectable Solution.
1 bottle of Powder For Injectable Solution equipped with BIO-SET
10 vials of Powder For Injectable Solution equipped with BIO-SET
Source Package Leaflet: AIFA (Italian Medicines Agency). Content published in January 2016. The information present may not be up-to-date.
To have access to the most up-to-date version, it is advisable to access the AIFA (Italian Medicines Agency) website. Disclaimer and useful information.
01.0 NAME OF THE MEDICINAL PRODUCT
CERNEVIT
02.0 QUALITATIVE AND QUANTITATIVE COMPOSITION
Each bottle of Powder for Solution for Injection contains:
03.0 PHARMACEUTICAL FORM
Powder for solution for injection.
04.0 CLINICAL INFORMATION
04.1 Therapeutic indications
Vitamin intake corresponding to the daily needs of adults and children over 11 years of age, in situations that require an injectable vitamin supplement when oral intake is contraindicated, impossible or insufficient (malnutrition, digestive malabsorption, parenteral nutrition )
04.2 Posology and method of administration
Adults and children over 11 years of age:
1 bottle per day
Particular posologies:
When an increased supply of nutrients is required (for example severe burns) CERNEVIT can be administered at daily doses 2-3 times higher than those normally used.
How to use:
Intravenously:
See section 6.6 "instructions for" use and handling ".
With a syringe, inject the contents of a solvent vial (5 ml of water for injections) into the bottle. Shake lightly to dissolve the lyophilisate. E.V. slow or by fusion in physiological solution, glucose, etc. CERNEVIT can enter the composition of nutritional mixtures in association with carbohydrates, lipids, amino acids, electrolytes, after verifying the compatibility and stability of each type of mixture.
Intramuscularly:
Dissolve the lyophilisate in 2.5 ml of solvent (water for injections), as mentioned above.
04.3 Contraindications
Known hypersensitivity to one of the components of this specialty; in particular, do not inject people with a history of intolerance to thiamine (vitamin B1).
Pregnancy.
04.4 Special warnings and appropriate precautions for use
Very high doses of vitamin A in animals are teratogenic and have been associated in isolated cases with malformations in humans. Daily amounts of vitamin A higher than 10,000 IU should be avoided during pregnancy (especially in the first months) and a doctor should be consulted for advice on the total amount of vitamin A that can be taken through the different sources.
Given the presence of vitamin A (retinol) in this specialty, take into account the doses administered in case of combinations with other preparations already containing this vitamin.
CERNEVIT does not contain vitamin K. The latter will need to be administered separately if needed.
After IV bolus injection, a moderate increase in SGPT transaminases only has been observed in some patients with progressive inflammatory enterocolitis; rapidly reversible increase on discontinuation of dosing. It is therefore recommended that transaminase levels be monitored in this type of patient.
Due to the presence of glycolic acid, repeated and prolonged administration in patients with jaundice of hepatic origin or important biological cholestasis requires careful monitoring of liver functions.
04.5 Interactions with other medicinal products and other forms of interaction
Vitamin B6 can antagonize the therapeutic effect of levo dopa.
04.6 Pregnancy and lactation
In the absence of experimental data, it is recommended not to administer the product in case of pregnancy or during lactation.
04.7 Effects on ability to drive and use machines
Not relevant.
04.8 Undesirable effects
Sporadic increase in SGPT transaminases after IV bolus injection in some subjects (see: "precautions for use").
Given the presence of vitamin B1, it is possible to observe anaphylactic reactions in subjects with latent allergy (see: "contraindications").
Possibility of pain at the intramuscular injection site. In this case, slow intravenous or deep intramuscular injection is recommended.
04.9 Overdose
Manifestations of hypervitaminosis A and hypervitaminosis D (symptoms linked to hypercalcemia) are possible in case of prolonged administration of large doses of these vitamins.
05.0 PHARMACOLOGICAL PROPERTIES
05.1 Pharmacodynamic properties
ATC: A11BA
CERNEVIT is a "balanced combination of all water-soluble and fat-soluble vitamins, necessary for the metabolism of adults and children over 11 years of age, with the exception of vitamin K.
This composition meets the recommendations of the AMA (American Medical Association) and has been approved by the FDA (Food and Drug Administration).
05.2 "Pharmacokinetic properties
The acute toxicity (LD 50) of CERNEVIT was studied in mice of both sexes.
Despite the very rapid injection rate, LD 50 was found to be 250 times higher than the dose expected in the human clinic, thus revealing a very high margin.
During the studies on the potential toxicity of CERNEVIT for repeated administration, conducted in the dog, for 30 days at doses 48 times higher than those expected in humans, no toxicity phenomena emerged, neither on the behavioral level, nor from the haematological and blood chemistry results. and from the histological examination of the light organs.
The solvents of the fat-soluble vitamins (glycolic acid + leticin) have been the subject of a comprehensive toxic-pharmacological study including reproduction, mutagenesis and carcinogenicity studies.
The results of these investigations have shown that these solvents can be safely administered intravenously and intramuscularly.
Local tolerability studies together with those for the evaluation of allergenic and anaphylactogenic potential have shown that CERNEVIT is well tolerated and does not induce allergy or anaphylaxis phenomena.
The good tolerability of CERNEVIT has also been confirmed in humans.
05.3 Preclinical safety data
-----
06.0 PHARMACEUTICAL INFORMATION
06.1 Excipients
Glycol;
glycolic acid;
soy leticin;
sodium hydroxide;
hydrochloric acid.
06.2 Incompatibility
No data are known in this regard.
06.3 Period of validity
3 years.
06.4 Special precautions for storage
Keep away from light and heat.
06.5 Nature of the immediate packaging and contents of the package
Powder for Injection Solution in brown glass bottle.
Box of 1 vial of Powder for Injectable Solution.
Powder for Injection Solution in brown glass bottle.
Box of 10 vials of Powder for Injectable Solution.
Powder for Injection Solution in a brown glass bottle equipped with BIO-SET.
Box of 1 bottle of Powder for Injectable Solution.
Powder for Injection Solution in a brown glass bottle equipped with BIO-SET.
Box of 10 vials of Powder for Injectable Solution.
06.6 Instructions for use and handling
Cernevit (bottle without BIO-SET)
With a syringe, inject the contents of a solvent vial (5 ml of water for injections) into the bottle. Shake lightly to dissolve the lyophilisate.
E.V. slow or by fusion in physiological solution, gucosate, etc.
Cernevit BIO-SET:
Cernevit BIO-SET allows reconstitution directly in bags equipped with an injection site (whether they are single or multi-compartment bags).
Single bag:
Remove the cap by twisting and pulling it to break the safety ring;
Connect the BIO-SET directly to the bag through the injection site;
Activate the BIO-SET by applying light pressure on the transparent mobile part of BIO-SET. This action pierces the rubber stopper of the bottle.
Place the combined system (Cernevit BIO-SET + the infusion bag) vertically with the bag on top. Squeeze the bag lightly several times to transfer the solution into the bottle (approximately 5 ml). Shake the bottle to reconstitute Cernevit.
Turn the combined system upside down by bringing the bag down. Lightly squeeze the infusion bag several times to push the air into the bottle headspace allowing the solution to be transferred into the bag.
Repeat steps 4 and 5 until the bottle is empty.
Remove and discard the Cernevit BIO-SET.
Multi-compartment bag:
Reconstitution of Cernevit BIO-SET must be done before activating the multi-compartment bag (before opening the non-permanent membranes and before mixing the contents of each compartment).
Place the multi-compartment bag on a work surface;
Remove the cap of Cernevit BIO-SET by turning and pulling it to break the safety ring;
Connect the BIO-SET directly to the multi-compartment bag through the injection site;
Activate the BIO-SET by applying light pressure on the transparent mobile part of BIO-SET. This action pierces the rubber stopper of the bottle;
Hold the bottle vertically. Squeeze the bag lightly several times to transfer the solution into the bottle (approximately 5 ml). Shake the bottle to reconstitute Cernevit.
Turn the combination system upside down while holding the bottle upside down. Lightly squeeze the infusion bag several times to push air into the headspace in the bottle thus allowing the solution to be transferred into the bag.
Repeat steps 5 and 6 until the bottle is empty.
Remove and discard the Cernevit BIO-SET.
Finally, activate the multi-compartment bag
Attention:
Make sure that the BIO-SET is always connected to the injection point during the whole process and reconstitution.
07.0 MARKETING AUTHORIZATION HOLDER
CLINTEC PARENTERAL
6, Avenue L. Pasteur - BP 56
78311 MAUREPAS CEDEX
FRANCE
Dealer for Italy:
BAXTER S.p.A ..
Viale Tiziano 25
00196 ROME
08.0 MARKETING AUTHORIZATION NUMBER
CERNEVIT - 1 bottle of Powder for Injectable Solution AIC N. 027959016
CERNEVIT - 10 vials of Powder for Injectable Solution AIC N. 027959028
CERNEVIT BIO - SEP
1 bottle of Powder for Injectable Solution AIC N. 027959030
CERNEVIT BIO - SEP
10 bottles of Powder for Injectable Solution AIC N. 027959042
09.0 DATE OF FIRST AUTHORIZATION OR RENEWAL OF THE AUTHORIZATION
CERNEVIT - 1 bottle of Powder for Injection 1/6/02
CERNEVIT - 10 vials of Powder for Injectable Solution June 2004
CERNEVIT BIO - SET - 1 bottle of Powder for Injectable Solution June 2004
CERNEVIT BIO - SET - 10 vials of Powder for Injectable Solution June 2004
10.0 DATE OF REVISION OF THE TEXT
01/06/2004