What is Lojuxta - lomitapide and what is it used for?
Lojuxta is a medicine that contains the active substance lomitapide, used to treat adult patients with homozygous familial hypercholesterolaemia, an inherited disease that causes blood levels of cholesterol (a type of fat) to rise. The medicine is used in conjunction with a low-fat diet and other medicines to reduce the level of fat in the blood. Whenever possible, the patient's disease should be diagnosed by genetic testing.
How is Lojuxta used - lomitapide?
Lojuxta can only be obtained with a prescription. It is available in capsule form (5, 10 and 20 mg) to be taken by mouth on an empty stomach, at least two hours after the evening meal. Therapy should be initiated and monitored by a physician experienced in the treatment of conditions resulting in increased blood fat levels. Treatment should start at a dose of 5 mg once daily; if well tolerated, the dosage can be gradually increased up to the maximum dose of 60 mg. Patients with impaired liver function or patients on kidney dialysis and those taking certain other medicines will need to take a reduced dosage. For more information, see the package leaflet.
How does Lojuxta - lomitapide work?
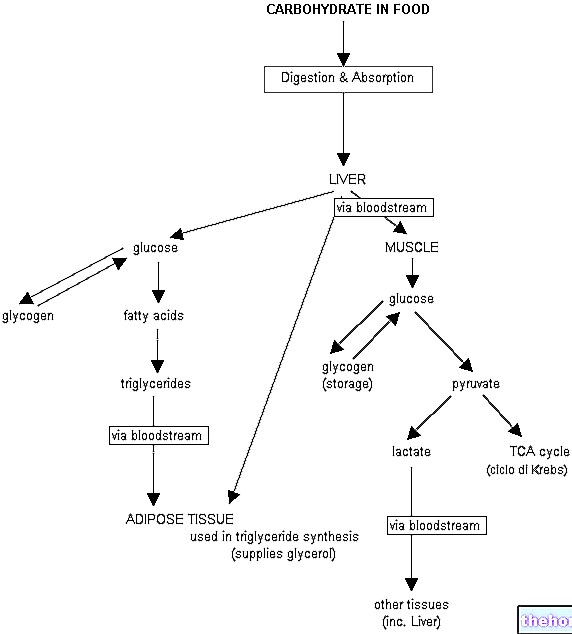
The active substance in Lojuxta, lomitapide, blocks the action of a substance in the body called 'microsomal triglyceride transfer protein', which is found in the liver cells and intestines. This substance is responsible for the accumulation of substances. fat such as cholesterol and triglycerides in larger particles called lipoproteins, which are subsequently released into the blood. By blocking this protein, Lojuxta decreases the level of fat released into the blood, thus helping to reduce the cholesterol rate in hypercholesterolemia.
What benefit has Lojuxta - lomitapide shown during the studies?
The benefits of Lojuxta in lowering blood cholesterol were evaluated in a main study involving 29 patients with homozygous familial hypercholesterolemia. All patients were treated with Lojuxta in combination with other fat-lowering medicines in the blood. Lojuxta was not compared with other therapies. The main measure of effectiveness was changes in blood levels of LDL cholesterol (low density lipoprotein), commonly known as "bad cholesterol", in patients 26 weeks after treatment. . On average, patients' LDL cholesterol levels were reduced by 40%.
What is the risk associated with Lojuxta - lomitapide?
The most serious side effect seen in some patients treated with Lojuxta is an abnormal increase in enzyme levels in the liver. The most common side effects are intestinal problems, which may affect up to 9 in 10 people: diarrhea, nausea, dyspepsia (heartburn stomach) and vomiting were seen in more than 3 in 10 people, while at least 2 in 10 people reported abdominal pain, discomfort and bloating, constipation and flatulence. For the full list of side effects reported with Lojuxta, see the package leaflet. . Lojuxta should not be used in pregnant women. It must also not be used in patients with moderate to severe liver impairment or with abnormal and unexplained liver test results, or in people with significant or long-term bowel problems. Lojuxta must not be used concomitantly with a dose of simvastatin (another medicine used to lower blood cholesterol levels) greater than 40 mg or with certain other medicines that affect the breakdown of lomitapide in the body. For a complete list of these. limitations, see package leaflet.
Why has Lojuxta - lomitapide been approved?
The Agency's Committee for Medicinal Products for Human Use (CHMP) decided that Lojuxta's benefits are greater than its risks and recommended that it be approved for use in the EU. The CHMP took into account that patients with Homozygous familial hypercholesterolemia with unmet medical demand found benefit in terms of lowering LDL cholesterol levels. However, the CHMP noted that the long-term benefit for the cardiovascular system has yet to be confirmed. The Committee also noted that Lojuxta produces side effects in the gut in most patients, sometimes of such a magnitude that discontinuation of therapy is necessary, and that the medicine has led to an increase in enzyme levels in the liver of which the long-term consequences are not known. Therefore, the committee has established that these effects must be closely monitored and managed. Lojuxta was authorized under "exceptional circumstances" because it was not possible to obtain complete information on Lojuxta due to the rarity of the disease. Each year the European Medicines Agency will review the new information available and this summary will be updated accordingly.
What information is still awaited for Lojuxta - lomitapide?
As Lojuxta has been authorized in exceptional circumstances, the company that markets the medicine will carry out a long-term study in patients treated with Lojuxta to provide further data on its safety and efficacy, including its side effects on the liver, stomach, intestines and cardiovascular system. The study will also provide data on the pregnancies of women taking the medicine and on compliance by healthcare professionals with recommendations to screen and monitor patients before and during therapy.
What measures are being taken to ensure the safe and effective use of Lojuxta - lomitapide?
A risk management plan has been developed to ensure that Lojuxta is used as safely as possible. Based on this plan, safety information has been added to the summary of product characteristics and package leaflet for Lojuxta, including the appropriate precautions to be followed by healthcare professionals and patients. In addition, the company that markets Lojuxta will provide all doctors who may prescribe Lojuxta with educational materials containing guidance on how to select suitable patients as well as basic safety information, including side effects, interactions with other medicines and use in older women. Information will also be provided for distribution to patients, including a leaflet and an alert card.
More information about Lojuxta - lomitapide
On 31 July 2013, the European Commission granted a "Marketing Authorization" for Lojuxta, valid throughout the European Union. For more information on therapy with Lojuxta, read the package leaflet (included with the EPAR) or consult your doctor. or the pharmacist. Last update of this summary: 08-2013
The information on Lojuxta - lomitapide published on this page may be out of date or incomplete. For a correct use of this information, see the Disclaimer and useful information page.