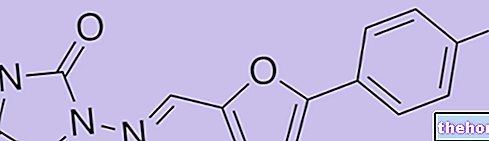
anthraquinone: among all the anthraquinone drugs, which exert the same laxative action (such as cassia, rhubarb and senna), aloe is undoubtedly the most powerful, given its highly irritating action. Its anthraquinones are so irritating that they can create real ulcers of the intestinal mucosa.
The laxative effect exerted by aloe is not, however, immediate, as it sets in after about 6-8 hours.
, cortisone and cardioactive glycosides (increased potassium loss with consequent hypokalaemia). Also, aloe juice can decrease the absorption of other medications.
There have been rare cases of dermatitis and allergy in predisposed subjects and in people allergic to Liliaceae.
, during the menstrual cycle, in children and in the presence of: varicose veins, hemorrhoids, kidney problems, inflammatory bowel disease, appendicitis and Crohn's disease.However, it should be noted that, to date (April 2021), with the entry into force of the new European regulation, the use of products based on aloe, or obtained from aloe, containing hydroxyanthracene derivatives responsible for the effect purgatives (such as aloe juice for the note) are prohibited due to the health risks they can induce.
Deepening: New European Regulation of 18 March 2021
On April 8, 2021, the ban on marketing foods and food supplements containing hydroxyanthracenes and their derivatives, a family of molecules contained in various plants, such as aloe, cassia, rhubarb and senna, came into force.
More in detail, the new European Regulation of March 18, 2021 - which came into force, precisely, April 8, 2021 - modifies Annex III of Regulation (EC) No. 1925/2006 of the European Parliament and of the Council to as regards the botanical species containing hydroxyanthracene derivatives.
The full text can be consulted by clicking here. However, we can summarize the main points as follows:
- The following are added to the list of substances whose use in food is prohibited (Annex III part A of the aforementioned regulation):
- Aloe-emodin and all preparations in which this substance is present;
- Emodin and all preparations in which this substance is present;
- Preparations based on leaves of Aloe species containing hydroxyanthracene derivatives;
- Dantrone and all preparations in which this substance is present.
- The following are added to the list of substances whose use in food is subject to Community surveillance (Annex III part C):
- Preparations based on the root or rhizome of Rheum palmatum L., Rheum officinale Baillon and their hybrids containing hydroxyanthracene derivatives;
- Preparations based on leaves or fruits of Cassia senna L. containing derivatives of hydroxyanthracene;
- Preparations based on bark of Rhamnus frangula L. o Rhamnus purshiana A.D. containing derivatives of hydroxyanthracene.




















.jpg)