Active ingredients: Permethrin
SCABIACID 5% cream 30 g tube
SCABIACID 5% cream 60 g tube
Why is Scabiacid used? What is it for?
PHARMACOTHERAPEUTIC CATEGORY
Pyrethroid ectoparasiticides
THERAPEUTIC INDICATIONS
Permethrin 5% cream is indicated for the treatment of scabies in adults and children over 2 months of age.
Contraindications When Scabiacid should not be used
Hypersensitivity to the active substance permethrin or to other substances belonging to the group of pyrethrins or pyrethroids or to any of the excipients listed in the Composition section. In these cases, treatment with a chemically different anti-scabies agent must be switched.
Generally contraindicated in pregnancy and during lactation.
Precautions for use What you need to know before taking Scabiacid
In case of hypersensitivity to chrysanthemums or other composites, the treatment should be administered only if strictly necessary.
When using SCABIACID 5%, care must be taken to prevent the cream from entering the eyes or coming into contact with mucous membranes (e.g. nasopharyngeal space, genital area) or with open wounds. In case of accidental contact, rinse immediately with water.
Pediatric population
There is only limited experience with SCABIACID in children aged 2 months to 23 months. Therefore, treatment should only be administered under close medical supervision in this age group.
For cutaneous use only!
If you apply the cream to other people, the use of gloves is recommended.
This medicinal product contains cetostearyl alcohol which can cause local skin reactions (e.g. contact dermatitis). SCABIACID 5% is harmful to all types of insects and also to aquatic life forms (fish, daphnia, algae). Avoid contamination of aquariums and terrariums.
The paraffin excipient can reduce the functioning and therefore the reliability of latex products (eg condoms, diaphragms) used at the same time.
KEEP OUT OF THE SIGHT AND REACH OF CHILDREN.
Interactions Which drugs or foods can modify the effect of Scabiacid
Treatment of eczematous-type reactions with corticosteroids should be evaluated prior to treatment with permethrin 5% cream, as there is a risk of exacerbation of scabies infestation due to reduced immune response to the mite. The probability of interactions between the two treatments leading to the enhancement of adverse reactions or reduction of efficacy is however low.
Warnings It is important to know that:
Use in pregnancy and during lactation
The data available to date on the use of permethrin 5% cream have not shown any risk to the fetus. The amount of permethrin absorbed systemically after applying the cream to the entire body surface is extremely low. However, permethrin can pass the placental barrier Negativity of mutagenicity tests and low mammalian toxicity suggest that treatment with permethrin carries minimal risk to the fetus.
It is shown that following oral administration, very low concentrations of permethrin are excreted in the milk of cows. It is not known whether permethrin is excreted in human milk. However, concentrations of permethrin in milk are unlikely to pose a risk to the newborn as only extremely small amounts of permethrin are absorbed systemically and in theory only a very small percentage can pass into human milk.
During pregnancy and breastfeeding, the medicine must be used in cases of real need and under direct medical supervision.
Dosage and method of use How to use Scabiacid: Dosage
SCABIACID should be applied to clean, dry and fresh skin. The product should not be applied immediately after taking a hot bath. Treatment of children under two years of age should only be carried out under medical supervision. Application of the cream by older children should be done under adult supervision.
Dosage
Unless otherwise directed by your doctor, the recommended dose is as follows:
Adults and adolescents over 12 years of age
Apply up to 30 g of cream (corresponding to a 30 g tube or ½ 60 g tube). Some adults may need an additional tube of cream to fully cover the body, but more than 60g (1 x 60g tube or 2 x 30g tube) should never be used for any single application.
Pediatric population
- Children from 6 to 12 years of age:
Apply up to 15 g of cream (corresponding to ½ of 30 g tube or ¼ of 60 g tube).
- Children from 2 months to 5 years of age:
Up to 7.5 g of cream (corresponding to ¼ of a 30 g tube or? Of a 60 g tube).
The safety and efficacy of SCABIACID in children aged less than 2 months have not been established. No data are available.
Method of administration
For cutaneous use only. The medicine should not be ingested.
Carefully apply a thin layer of cream to the skin (for cutaneous use).
Adults should apply the cream evenly all over the body including the neck, palms and soles of the feet. The head and face can be avoided if there is no scabies efflorescence in this region.
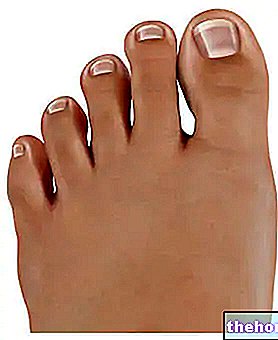
During application, the areas between the fingers and toes (including the fingers and under the toenails), wrists, elbows, armpits, external genitals, breasts and buttocks should be treated with special care.
Pediatric population
Children should apply the cream evenly all over the body, including the palms of the hands, soles of the feet, neck, face, ears and scalp. Parts of the skin around the mouth (as the cream could be removed with the tongue) and around the eyes should be avoided. Children must be supervised to ensure that they do not lick the cream from their hands. If necessary, children should wear gloves.
There is only limited experience with SCABIACID in children aged 2 months to 23 months. Therefore, treatment should only be administered under close medical supervision in this age group.
Senior citizens
Elderly patients (over 65 years of age) should use the cream in the same way as in adults, but in addition, the face, ears and scalp should be treated. Care should be taken to avoid applying the cream on the areas of skin around the eyes.
The cream should be left to act for at least 8 hours. If you wash your hands before 8 hours have passed from applying the cream, you need to reapply it on them.
Wash the body thoroughly 8-12 hours after applying the cream.
In 90% of cases a single application of SCABIACID is sufficient to eliminate the infestation. If the lesions do not show signs of healing or new lesions appear, a second application of the cream can be used after 7 days.
Overdose What to do if you have taken too much Scabiacid
There are no reports of overdose of permethrin 5% cream.
Excessive application of cream to the skin could lead to local adverse reactions. The application of a whole tube (30 g) of cream on a two month old baby would correspond to a dose of about 350 mg / kg of body weight.
Even if fully absorbed, this dose is unlikely to cause clear signs of systemic toxicity.
METHOD "D" INTERVENTION IN CASE OF EXCESSIVE DOSE
In case of accidental ingestion of the product by a child, gastric lavage could be used, to be carried out within 2 hours of ingestion.
Treatment of hypersensitivity reactions should be symptomatic.
Side Effects What are the side effects of Scabiacid
Like all medicines, SCABIACID can cause side effects, although not everybody gets them.
Cases of
- short-lived burning or stinging irritation. This is usually in a mild form and happens more frequently in patients with severe scabies.
- Transient signs and symptoms of skin irritation such as rash or itching, erythema, edema, eczema.
Undesirable effects are grouped on the basis of system organ class. Within each system organ class, the frequency is defined as follows: common (≥ 1/100,
Itching can continue for up to 4 weeks after treatment and is due to an allergic reaction to dead scabies mites. This is normal and not a sign that the treatment has not been effective.
Compliance with the instructions contained in the package leaflet reduces the risk of undesirable effects.
Reporting of side effects
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the national reporting system at www.agenziafarmaco.it/it/responsabili. By reporting side effects you can help provide more information on the safety of this medicine.
Expiry and Retention
See the expiration date indicated on the package. This date refers to the product in intact and correctly stored packaging.
ATTENTION: do not use the product beyond the expiration date indicated on the package.
Do not store above 30 ° C, do not freeze.
Other information
COMPOSITION
Each 30g tube of 5% cream contains:
Active ingredient: Permethrin 5% (cis isomers: trans 25:75) 1.5 g.
Each 60g tube of 5% cream contains:
Active ingredient: Permethrin 5% (cis isomers: trans 25:75) 3 g.
Excipients: Cetostearyl alcohol, paraffin, cetomacrogol 1000, isopropyl myristate, benzyl alcohol, purified water.
PHARMACEUTICAL FORM AND CONTENT
Cream 5% w / w cream - 30 g and 60 g tube.
Source Package Leaflet: AIFA (Italian Medicines Agency). Content published in January 2016. The information present may not be up-to-date.
To have access to the most up-to-date version, it is advisable to access the AIFA (Italian Medicines Agency) website. Disclaimer and useful information.
01.0 NAME OF THE MEDICINAL PRODUCT
SCABIACID 5% CREAM
02.0 QUALITATIVE AND QUANTITATIVE COMPOSITION
Permethrin 5% (isomers cis: trans 25:75).
For excipients see section 6.1.
03.0 PHARMACEUTICAL FORM
Cream for dermatological use.
04.0 CLINICAL INFORMATION
04.1 Therapeutic indications
Permethrin 5% cream is indicated for the treatment of scabies in adults and children over 2 months of age.
04.2 Posology and method of administration
SCABIACID should be applied to clean, dry and fresh skin. The product should not be applied immediately after taking a hot bath.
Treatment of children under two years of age should only be carried out under medical supervision. Application of the cream by older children should be done under adult supervision.
Dosage
Unless otherwise directed by your doctor, the recommended dose is as follows:
Adults and adolescents over 12 years of age:
Apply up to 30 g of cream (corresponding to a 30 g tube or ½ 60 g tube).
Some adults may need an additional tube of cream to fully cover the body, but more than 60g (1 x 60g tube or 2 x 30g tube) should never be used for any single application.
Pediatric population
Children from 6 to 12 years of age:
Apply up to 15 g of cream (corresponding to ½ of 30 g tube or ¼ of 60 g tube).
Children from 2 months to 5 years of age:
Up to 7.5 g of cream (corresponding to ¼ of a 30 g tube or 1/8 of a 60 g tube).
The safety and efficacy of SCABIACID in children aged less than 2 months have not been established. No data are available.
Method of administration
For cutaneous use only. The medicine should not be ingested.
Carefully apply a thin layer of cream to the skin (for cutaneous use).
Adults should apply the cream evenly all over the body including the neck, palms and soles of the feet. The head and face can be avoided if there is no scabies efflorescence in this region.
During application, the areas between the fingers and toes (including the fingers and under the toenails), wrists, elbows, armpits, external genitals, breasts and buttocks should be treated with special care.
Pediatric population
Children should apply the cream evenly all over the body, including the palms of the hands, soles of the feet, neck, face, ears and scalp. Parts of the skin around the mouth (as the cream could be removed with the tongue) and around the eyes should be avoided. Children must be supervised to ensure that they do not lick the cream from their hands. If necessary, children should wear gloves.
There is only limited experience with SCABIACID in children aged 2 months to 23 months. Therefore, treatment should only be administered under close medical supervision in this age group.
Senior citizens:
Elderly patients (over 65 years of age) should use the cream in the same way as in adults, but in addition, the face, ears and scalp should be treated. Care should be taken to avoid applying the cream on the areas of skin around the eyes.
The cream should be left to act for at least 8 hours. If you wash your hands before 8 hours have passed from applying the cream, you need to reapply it on them.
Wash the body thoroughly 8-12 hours after applying the cream.
In 90% of cases a single application of SCABIACID is sufficient to eliminate the infestation. If the lesions do not show signs of healing or new lesions appear, a second application of the cream can be used after 7 days.
04.3 Contraindications
Hypersensitivity to the active substance permethrin or to other substances belonging to the group of pyrethrins or pyrethroids or to any of the excipients listed in section 6.1. In these cases, treatment with a chemically different anti-scabies agent must be switched.
Generally contraindicated in pregnancy and during lactation.
04.4 Special warnings and appropriate precautions for use
In case of hypersensitivity to chrysanthemums or other composites, the treatment should be administered only if strictly necessary.
When using SCABIACID 5%, care must be taken to prevent the cream from entering the eyes or coming into contact with mucous membranes (e.g. nasopharyngeal space, genital area) or with open wounds.
In case of accidental contact, rinse immediately with water.
Pediatric population
There is only limited experience with SCABIACID in children aged 2 months to 23 months. Therefore, treatment should only be administered under close medical supervision in this age group.
For cutaneous use only!
If you apply the cream to other people, the use of gloves is recommended.
This medicinal product contains cetostearyl alcohol which can cause local skin reactions (e.g. contact dermatitis).
SCABIACID 5% is harmful to all types of insects and also to aquatic life forms (fish, daphnia, algae). Avoid contamination of aquariums and terrariums.
The paraffin excipient can reduce the functioning and therefore the reliability of latex products (eg condoms, diaphragms) used at the same time.
04.5 Interactions with other medicinal products and other forms of interaction
Treatment of eczematous-type reactions with corticosteroids should be evaluated prior to treatment with permethrin 5% cream, as there is a risk of exacerbation of scabies infestation due to reduced immune response to the mite. The probability of interactions between the two treatments leading to the enhancement of adverse reactions or reduction of efficacy is however low.
04.6 Pregnancy and breastfeeding
The data available to date on the use of permethrin 5% cream have not shown any risk to the fetus. The amount of permethrin absorbed systemically after applying the cream to the entire body surface is extremely low. However, permethrin can pass the placental barrier Negativity of mutagenicity tests and low mammalian toxicity suggest that treatment with permethrin carries minimal risk to the fetus.
It has been shown that following oral administration, very low concentrations of permethrin are excreted in the milk of cows. It is not known whether permethrin is excreted in human milk. However, permethrin concentrations in milk are unlikely to pose a risk to the infant as only extremely small amounts of permethrin are absorbed systemically and in theory only a very small percentage can pass into human milk.
During pregnancy and breastfeeding, the medicine must be used in cases of real need and under direct medical supervision.
04.7 Effects on ability to drive and use machines
None known.
04.8 Undesirable effects
Cases of
- short-lasting burning or urticating irritation. This is usually in a mild form and happens more frequently in patients with severe scabies.
- transient signs and symptoms of skin irritation such as rash or itching, erythema, edema, eczema.
Undesirable effects are grouped on the basis of system organ class. Within each system organ class, frequency is defined as follows: common (≥1 / 100,
Itching can continue for up to 4 weeks after treatment and is due to an allergic reaction to dead scabies mites. This is normal and not a sign that the treatment has not been effective.
Reporting of suspected adverse reactions
Reporting of suspected adverse reactions occurring after authorization of the medicinal product is important as it allows continuous monitoring of the benefit / risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system. "address www.agenziafarmaco.gov.it/it/responsabili.
04.9 Overdose
There are no reports of overdose of permethrin 5% cream.
Excessive application of cream to the skin could lead to local adverse reactions. Application of a full tube (30 g) of cream to a two month old baby would correspond to a dose of approximately 350 mg / kg body weight. Even if fully absorbed, this dose is unlikely to cause clear signs of toxicity. systemic.
In case of accidental ingestion of the product by a child, gastric lavage could be used, to be carried out within 2 hours of ingestion.
Treatment of hypersensitivity reactions should be symptomatic.
05.0 PHARMACOLOGICAL PROPERTIES
05.1 Pharmacodynamic properties
Pharmacotherapeutic group: pyrethroid ectoparasiticides.
ATC code: P03AC04.
The main physiological action in parasites exposed to permethrin is the induction of electrochemical abnormalities across excitable cell membranes, leading to sensory hyperexcitability, incoordination and prostration.
Pediatric population
Newborns and infants:
The safety and efficacy of permethrin in newborns and infants under 2 months of age have not been established as no data from prospective studies or larger series are available. A limited number of case reports in the treatment of younger children at 2 months with scabies do not suggest particular safety concerns for the use of topical permethrin in this group, however no definitive conclusion can be drawn.
05.2 Pharmacokinetic properties
In mammals, permethrin is rapidly metabolised via the hydrolysis of esters into inactivated metabolites which are excreted primarily via the kidneys. The major metabolites are detectable in the urine within 7 hours of applying the cream to the whole body of healthy volunteers or affected patients. The highest levels of excretion are detectable within 48 hours, but in some individuals very low levels of metabolites may be found in the urine as long as 28 days after treatment. The overall pattern of excretion indicates that approximately 0.5% of the applied permethrin is absorbed in the first 48 hours.
Investigations conducted with 5% cream in humans revealed a mean percutaneous absorption rate of 0.47 ± 0.3% in healthy subjects and 0.52 ± 0.3% in patients.
Pharmacokinetic properties have only been studied in adult subjects (6 healthy volunteers and 6 patients with scabies).
The absorbed permethrin is rapidly broken down by esterases as well as by hydrolases. After oral administration, peak plasma concentrations are reached in approximately 4 hours. The mixture of isomers is excreted in the urine in the form of glucuronides, sulphates etc. as CI2CA cis-trans [(3- (2,2-dichlorovinyl) -2,2-dimethylcyclopropane-1-carboxylic acid)] and after the oxidation of 3 PBA (3-phenoxybenzoic acid) After oral administration, up to 6% is excreted unchanged in the faeces, while with cutaneous administration, unchanged permethrin is practically undetectable.
05.3 Preclinical safety data
In vivo and in vitro genetic toxicity studies were all negative, revealing that permethrin cannot induce mutagenic changes.
There is no evidence from acute and chronic toxicity studies to indicate the presence of previously unknown adverse effects in humans. Furthermore, there is no relevant evidence for genotoxic or carcinogenic potential. Long-term studies in rats did not reveal any oncogenic effects. Similar studies in mice showed a species-specific increase in the incidence of pulmonary edema at high doses and in female mice only, when permethrin was administered with food for two years (approximately 750 mg / kg of body weight / day). These results are not believed to indicate a potential oncogenicity of permethrin in humans.
In reproductive toxicity studies in mice, rats and rabbits after repeated oral administration of permethrin effects were observed only for doses well above the expected exposure for topical use of 5% cream. Following the intended use of this active substance, a serious adverse effect on aquatic (daphnia and fish) and terrestrial (plants) organisms is expected after passing through waste water.
06.0 PHARMACEUTICAL INFORMATION
06.1 Excipients
Cetostearyl alcohol, paraffin, cetomacrogol 1000, isopropyl myristate, benzyl alcohol, purified water.
06.2 Incompatibility
None known.
06.3 Period of validity
2 years.
06.4 Special precautions for storage
Do not store above 30 ° C, do not freeze.
06.5 Nature of the immediate packaging and contents of the package
Aluminum tube with polypropylene cap. The tube contains 30 g or 60 g of cream.
06.6 Instructions for use and handling
Once the package has been opened, use all the contents.
07.0 MARKETING AUTHORIZATION HOLDER
Valetudo Srl - Via Ghiaie, 6 - 24030 Presezzo (Bg)
08.0 MARKETING AUTHORIZATION NUMBER
SCABIACID 5% cream, 1 tube of 30 g AIC n. 036290017
SCABIACID 5% cream, 1 tube of 60 g AIC n. 036290031
09.0 DATE OF FIRST AUTHORIZATION OR RENEWAL OF THE AUTHORIZATION
July 8, 2006
10.0 DATE OF REVISION OF THE TEXT
March 2014