Active ingredients: Glatiramer (Glatiramer acetate)
Copaxone 20 mg / ml solution for injection, pre-filled syringe
Copaxone package inserts are available for pack sizes:- Copaxone 20 mg / ml solution for injection, pre-filled syringe
- Copaxone 40 mg / ml solution for injection, pre-filled syringe
Why is Copaxone used? What is it for?
Copaxone 20 mg / ml is a medicine that changes the way your body's immune system works (it is classified as an immunomodulatory agent). The symptoms of multiple sclerosis (MS) are thought to be caused by a defect in the body's immune system, which produces foci of inflammation in the brain and spinal cord.
Copaxone 20 mg / ml is used to reduce the number of times you have MS attacks (relapses). You have not been shown to help if you have a form of MS that has almost no relapse. Copaxone 20 mg / ml may have no effect on the duration of an MS attack or the intensity of pain during an attack.
It is used to treat patients who are unable to walk unaided.
Copaxone can also be used in patients who have experienced symptoms for the first time indicating a high risk of developing MS. Before you take this drug, your doctor will rule out any other reasons that might explain these symptoms.
Contraindications When Copaxone should not be used
Do not use Copaxone 20 mg / ml
- if you are allergic to glatiramer acetate or any of the other ingredients of this medicine
- if you are pregnant.
Precautions for use What you need to know before taking Copaxone
Talk to your doctor or pharmacist before using Copaxone 20 mg / ml
- if you have any kidney or heart problems as you may need regular tests and checks.
Children
Copaxone should not be used in children under 12 years of age.
Senior citizens
Copaxone has not been specifically studied in the elderly. Ask your doctor for advice on this.
Interactions Which drugs or foods may change the effect of Copaxone
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines.
Warnings It is important to know that:
Pregnancy and breastfeeding
Do not use Copaxone 20 mg / ml if you are pregnant. Tell your doctor if you become pregnant while using this medicine or if you are planning to become pregnant.
You should use an "effective contraceptive measure (for example the" pill "or condom) to avoid becoming pregnant while being treated with Copaxone.
Discuss with your doctor first if you wish to breastfeed while using Copaxone.
Driving and using machines
The influence of Copaxone 20 mg / ml on the ability to drive and use machines is not known.
Dose, Method and Time of Administration How to use Copaxone: Posology
Always use this medicine exactly as your doctor has told you. If in doubt, consult your doctor or pharmacist.
The daily dose in adults and adolescents aged 12 years or older is one pre-filled syringe (20 mg glatiramer acetate), administered under the skin (subcutaneously).
It is very important to inject Copaxone 20 mg / ml correctly
- In the tissue under the skin (subcutaneous tissue) only (see "Instructions for Use" below).
- At the dose indicated by the doctor. Only take the dose prescribed by your doctor.
- Never use the same syringe more than once. Unused product or any waste must be discarded.
- Do not mix or administer the contents of Copaxone 20 mg / ml pre-filled syringes with any other product.
- Do not use the solution if it contains particles. Use a new syringe.
The first time you use Copaxone 20 mg / ml you will be given full instructions and will be supervised by a doctor or healthcare professional. They will be with you while you give yourself the injection and for 30 minutes to make sure you have no problems.
Instructions for Use
Read these instructions carefully before using Copaxone 20 mg / ml.
Before the injection, make sure you have everything you need:
- One blister with one Copaxone 20 mg / ml pre-filled syringe
- A container for disposing of used needles and syringes.
- For each injection, take only one blister with one pre-filled syringe from the pack. Keep all remaining syringes in the carton.
- If the syringe has been stored in the refrigerator, take out the blister pack containing the syringe for at least 20 minutes before giving yourself the injection to warm it to room temperature.
Wash your hands thoroughly using soap and water.
If you wish to use the COPAXONE injection device to inject, please refer to the instructions for use supplied with the COPAXONE injection device.
Choose the injection site using the diagrams in Figure 1.
There are seven possible injection areas in your body: arms, thighs, hips and abdomen (belly). There are several injection sites in each injection area. Choose a different injection site each day. This reduces the possibility of any irritation or pain at the injection site. Rotate the injection sites within the same area. You don't use the same venue every time.
Note: Do not inject into a sore or discolored area or where you feel hardened masses or knots.
It is recommended that you keep a chart of the planned rotation of the injection sites and write it down on a diary. There are some places in your body where it may be difficult to inject yourself (such as the back of the arm). If you want to use these locations, you may need help.
How to give yourself the injection:
- Remove the syringe from its protective blister by removing the paper label.
- Remove the cap from the needle.
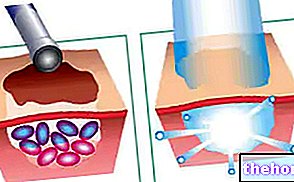
- Gently squeeze the skin with the thumb and forefinger of your free hand (Figure 2).
- Push the needle into the skin as shown in Figure 3.
- Inject the medicine by pushing the plunger down smoothly until the syringe is empty.
- Pull the syringe and needle straight out.
- Discard the syringe in a safe disposal container. Do not put the syringe in household waste, but carefully place it in a puncture-proof container as advised by your doctor or healthcare professional.
Overdose What to do if you have taken too much Copaxone
If you use more than one Copaxone 20 mg / ml syringe per day
Tell your doctor immediately.
If you forget to use Copaxone 20 mg / ml
Take it as soon as you remember, but do not take a double dose to make up for a forgotten dose. Take the next dose 24 hours later.
If you stop taking Copaxone 20 mg / ml
Do not stop using Copaxone 20 mg / ml without consulting your doctor.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
Side Effects What are the side effects of Copaxone
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Allergic reactions (hypersensitivity)
You may rarely develop a severe allergic reaction to this medicine.
Stop using Copaxone 20 mg / ml and contact your doctor immediately or go to the nearest hospital emergency department if you notice any of these side effects:
- rash (red spots or hives)
- swelling of the eyelids, face or lips
- sudden shortness of breath
- convulsions (fits)
- fainting
Other reactions after injection (immediate reaction after injection)
It is not common but some people may have one or more of the following symptoms in the minutes following the injection of Copaxone 20 mg / ml. They do not normally cause problems and usually go away within 30 minutes.
However, if the following symptoms last longer than 30 minutes, contact your doctor immediately or go to the emergency room of the nearest hospital:
- flushing (redness) in the chest or face (vasodilation)
- shortness of breath (dyspnoea)
- chest pain
- pounding and rapid heartbeat (palpitations, tachycardia)
The following side effects have been reported with Copaxone:
Very common (may affect more than 1 in 10 people):
- infections, flu
- anxiety, depression
- headache
- nausea
- skin rashes
- pain in the joints or back
- feeling faint, skin reactions at the injection site, including skin redness, pain, swelling, itching, tissue swelling, inflammation and hypersensitivity (these injection site reactions are not unusual and usually subside over time), pain not specific
Common (may affect up to 1 in 10 people):
- inflammation of the respiratory tract, gastroenteritis, herpes, inflammation of the ears, runny nose, dental abscess, vaginal thrush
- non-malignant skin growth (non-malignant skin neoplasm), tissue growth (neoplasm)
- swelling of the lymph nodes
- allergic reactions
- loss of appetite, weight gain
- nervousness
- altered taste, increased tightness of muscle tone, headache, speech problems, fainting, tremor
- double vision, eye problems
- ear problems
- cough, hay fever
- disorders of the anus or rectum, constipation, dental caries, indigestion, difficulty swallowing, fecal incontinence, vomiting
- abnormal liver function tests
- bruising, excessive sweating, itching, skin changes, hives
- neck pain
- urgent need to empty your bladder, frequent need to urinate, inability to empty your bladder properly
- chills, swelling of the face, loss of tissue under the skin at the injection site, local reactions, peripheral swelling caused by fluid accumulation, fever
Uncommon (may affect up to 1 in 100 people):
- abscess, infections of the skin and underlying soft tissues, boils, St. Anthony's fire, inflammation of the kidneys
- skin cancer
- increased white blood cell count, decreased white blood cell count, enlarged spleen, decreased platelet count, change in the shape of white blood cells
- enlarged thyroid, overactive thyroid
- low alcohol tolerance, gout, increased blood fat levels, increased blood sodium, decreased serum ferritin
- abnormal dreams, confusion, euphoric mood, seeing, hearing, smelling, tasting or feeling things that are not there (hallucinations), aggression, unusually high mood, personality disorder, suicide attempt
- falling asleep and pain in the hand (carpal tunnel syndrome), mental disorders, fits (convulsions), difficulty in writing and reading, muscle disorders, difficulty moving, involuntary muscle contractions, inflammation of the nerves, abnormal neuromuscular connection leading to a function abnormal muscle, involuntary rapid eye movement, paralysis, foot drop (peroneal nerve palsy), unconsciousness (stupor), blind spot vision
- cataract, eye damage to the cornea, dry eye, bleeding in the eye, drooping of the upper eyelid, pupil enlargement, weakening of the optic nerve leading to vision problems
- extrasystoles, slow heartbeats, episodic fast heartbeats
- varicose veins
- periodic stops in breathing, nosebleeds, abnormally fast or deep breathing (hyperventilation), feeling of narrowing of the throat, lung problems, inability to breathe due to narrowing of the throat (choking sensation)
- inflammation of the intestine, colon polyps, inflammation of the small intestine, belching, ulcer in the esophagus, inflammation of the gums, rectal bleeding, enlarged salivary glands
- gallstones, liver enlargement
- swelling of the skin and soft tissues, contact rash, red, painful skin thickening, skin thickening
- joint swelling, inflammation and pain (arthritis or osteoarthritis), inflammation and pain of the fluid pad lining the joints (present in some joints), flank pain, decreased muscle mass
- blood in urine, kidney stones, urinary tract disorders, urine abnormality
- abortion
- swollen breasts, difficulty getting an erection, falling down or out of the pelvic organs (pelvic prolapse), prolonged erection, prostate disorder, abnormal pap smear (abnormal cervical smear), testicular disorder, vaginal bleeding, disorders of the vagina
- cyst, hangover-like effects, low body temperature (hypothermia), non-specific inflammation, tissue destruction at the injection site, mucous membrane problems
- disorders after vaccination
Reporting of side effects
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet.
You can also report side effects directly via the national reporting system at: https://www.aifa.gov.it/content/segnalazioni-reazioni-avverse.
By reporting side effects you can help provide more information on the safety of this medicine.
Expiry and Retention
Keep this medicine out of the sight and reach of children
Store in a refrigerator (2 ° C - 8 ° C).
Copaxone 20 mg / ml pre-filled syringes can be kept out of the refrigerator at room temperature for one month. This can only be done once.After one month, Copaxone 20 mg / ml pre-filled syringes that have not been used and are still in their original packaging should be placed in the refrigerator.
Do not freeze.
Keep the pre-filled syringes in the outer carton to protect the medicine from light.
Do not use this medicine after the expiry date which is stated on the label and carton (EXP). The expiry date refers to the last day of that month.
Discard any syringe that contains particles.
Do not throw any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. This will help protect the environment.
What Copaxone 20 mg / ml contains
- The active ingredient is glatiramer acetate. 1 ml of solution for injection (the contents of one pre-filled syringe) contains 20 mg of glatiramer acetate.
- The other ingredients are mannitol and water for injections.
Description of what Copaxone 20 mg / ml looks like and contents of the pack
Copaxone 20 mg / ml solution for injection, pre-filled syringe is a sterile, clear solution free from visible particles.
Throw away the syringe if it contains particles and start again. Use a new syringe.
Copaxone is available in packs containing 7, 28 or 30 pre-filled syringes of 1 ml solution for injection or in a multipack containing 3 packs each of 30 pre-filled syringes of 1 ml solution for injection.
Not all pack sizes may be marketed
Source Package Leaflet: AIFA (Italian Medicines Agency). Content published in January 2016. The information present may not be up-to-date.
To have access to the most up-to-date version, it is advisable to access the AIFA (Italian Medicines Agency) website. Disclaimer and useful information.
01.0 NAME OF THE MEDICINAL PRODUCT
COPAXONE 20 MG / ML SOLUTION FOR INJECTION, PRE-FILLED SYRINGE
02.0 QUALITATIVE AND QUANTITATIVE COMPOSITION
1 ml of solution for injection contains 20 mg of glatiramer acetate *, equivalent to 18 mg of glatiramer base, for each pre-filled syringe.
* Glatiramer acetate is the acetate salt of synthetic polypeptides containing four natural amino acids: L-glutamic acid, L-alanine, L? Tyrosine and L-lysine, in the mole fraction ranges of 0.129-0.153, 0.392-0.462, 0.086-0.100 respectively and 0.300-0.374. The average molecular weight of glatiramer acetate is between 5,000-9,000 daltons.
For the full list of excipients, see section 6.1.
03.0 PHARMACEUTICAL FORM
Solution for injection, pre-filled syringe.
Clear solution free from visible particles.
04.0 CLINICAL INFORMATION
04.1 Therapeutic indications
Copaxone is indicated for the treatment of patients who have experienced a well-defined first clinical episode and who are considered to be at high risk of developing clinically defined multiple sclerosis (CDMS) (see section 5.1).
Copaxone is indicated to reduce the frequency of relapses in ambulatory patients (i.e. able to walk unaided) with relapsing multiple sclerosis (MS) with stages of remission. In clinical trials this was characterized by at least two bouts of neurological dysfunction in the preceding two-year period (see section 5.1).
Copaxone is not indicated in patients with primary or secondary progressive MS.
04.2 Posology and method of administration
Dosage
The recommended posology in adults is 20 mg glatiramer acetate (one pre-filled syringe), administered subcutaneously once daily.
It is currently unknown how long the patient should be treated.
A decision regarding long-term treatment must be made on a case-by-case basis by the attending physician.
Pediatric population
Children and adolescents: No prospective, controlled, randomized clinical studies or pharmacokinetic studies have been conducted in children or adolescents. However, the limited published data suggest that the safety profile in adolescents 12 to 18 years receiving Copaxone 20 mg per day subcutaneously is similar to that seen in adults. As there is not enough information available on the use of Copaxone in children under 12 years of age, no recommendation for use can be made. Consequently, Copaxone should not be used in this population.
Elderly patients
Copaxone has not been specifically studied in the elderly.
Patients with impaired renal function
Copaxone has not been specifically studied in patients with renal impairment (see section 4.4).
Method of administration
Patients should be instructed in self-injection techniques and should be followed by a healthcare professional the first time they inject the product themselves and for 30 minutes thereafter.
A different injection site should be chosen each day to reduce the possibility of any irritation or pain at the injection site. Self-injection sites include the abdomen, arms, hips and thighs.
04.3 Contraindications
Copaxone is contraindicated in the following conditions:
• Hypersensitivity to glatiramer acetate or mannitol.
• Pregnant women.
04.4 Special warnings and appropriate precautions for use
Copaxone should only be administered subcutaneously. Copaxone must not be administered intravenously or intramuscularly.
The initiation of Copaxone treatment should be supervised by a neurologist or physician experienced in the treatment of MS.
The treating physician should explain to the patient that a reaction associated with at least one of the following symptoms may occur within a few minutes after the injection of Copaxone: vasodilation (flushing), chest pain, dyspnoea, palpitations or tachycardia. Most of these symptoms is short-lived and resolves spontaneously without leaving any consequences. If a serious adverse event occurs, the patient should immediately discontinue treatment with Copaxone and contact the treating physician or an emergency room physician. Symptomatic treatment may be established at the discretion of the physician.
There is no evidence to suggest that any particular patient group is at special risk from these reactions. However, caution should be exercised when administering Copaxone to patients with pre-existing heart conditions. These patients should be followed up regularly during treatment.
Seizures and / or anaphylactoid or allergic reactions have been reported rarely.
Serious hypersensitivity reactions (e.g. bronchospasm, anaphylaxis or urticaria) may occur rarely. If reactions are severe, appropriate treatment should be instituted and Copaxone discontinued.
Reactive antibodies against glatiramer acetate have been recognized in patient sera during chronic daily treatment with Copaxone. Maximum levels were reached after an average treatment duration of 3-4 months and subsequently decreased and stabilized at a level slightly above baseline.
There is no evidence to suggest that these reactive antibodies against glatiramer acetate are neutralizing or that their formation is likely to affect the clinical efficacy of Copaxone.
In patients with renal impairment, renal function should be monitored during treatment with Copaxone. Although there is no evidence of glomerular deposition of immune complexes in patients, this possibility cannot be ruled out.
04.5 Interactions with other medicinal products and other forms of interaction
Interactions between Copaxone and other drugs have not been formally evaluated.
Interaction data with interferon beta are not available.
An "increased incidence of injection site reactions" has been observed in Copaxone-treated patients receiving concomitant administration of corticosteroids.
A study in vitro suggests that circulating glatiramer acetate is highly bound to plasma proteins, but that it is not moved by phenytoin or carbamazepine and that it does not itself move phenytoin or carbamazepine. However, since Copaxone theoretically has the potential to alter the distribution of protein bound substances, concomitant use of such medicinal products should be carefully monitored.
04.6 Pregnancy and lactation
Pregnancy
There are insufficient data from the use of glatiramer acetate in pregnant women. Animal studies are insufficient to demonstrate effects on pregnancy, embryo-fetal development, parturition and postnatal development (see section 5.3). they know the potential risks to man. Copaxone is contraindicated during pregnancy.
While using this product, a contraceptive measure should be considered.
Feeding time
No data are available regarding the excretion of glatiramer acetate, its metabolites or antibodies in human breast milk. Caution should be used when administering Copaxone to breastfeeding mothers. The relative risks and benefits to mother and child should be considered. .
04.7 Effects on ability to drive and use machines
Studies on the effects of the drug on the ability to drive and use machines have not been performed.
04.8 Undesirable effects
In all clinical studies, the most frequently observed adverse reactions were injection site reactions that were reported by the majority of patients treated with Copaxone. In controlled studies, the percentage of patients who reported these reactions at least once. was higher after Copaxone treatment (70%) than after placebo injections (37%). The more frequently reported injection site reactions, reported more frequently in Copaxone-treated patients than in patients treated with placebo, were: erythema, pain, presence of masses, pruritus, edema, inflammation and hypersensitivity.
A reaction associated with at least one or more of the following symptoms has been described as an immediate reaction after injection: vasodilation, chest pain, dyspnoea, palpitations or tachycardia. This reaction may occur within minutes after the Copaxone injection. At least one component of this immediate reaction after injection was reported at least once by 31% of patients treated with Copaxone compared with 13% in the placebo group.
All adverse reactions reported more frequently in Copaxone-treated patients than in placebo-treated patients are presented in the table below. These data are derived from four pivotal, double-blind, placebo-controlled clinical trials performed in a total of 512 patients treated with Copaxone and 509 patients treated with placebo for up to 36 months. Three studies included a total of 269 patients with relapsing-remitting multiple sclerosis (RRMS) treated with Copaxone and 271 patients treated with placebo for up to 35 months. The fourth study, performed in patients who experienced a first clinical episode and who were considered to be at high risk of developing clinically defined MS, included 243 patients treated with Copaxone and 238 patients treated with placebo for up to 36 months.
* incidence greater than 2% (> 2/100) in the Copaxone group compared to the placebo group. Undesirable effects without the * symbol report an "incidence of less than or equal to 2%.
§ The term "injection site reactions" (various types) includes all side effects seen at the injection site except injection site atrophy and necrosis which are listed separately in the table.
? includes terms related to injection site lipoatrophy.
In the fourth study described above, an open-label treatment phase followed the placebo control period (see section 5.1). No changes in the known Copaxone risk profile were observed during the open-label follow-up period of up to 5 years.
Rare reports (> 1/10000,
Reporting of suspected adverse reactions
Reporting of suspected adverse reactions occurring after authorization of the medicinal product is important as it allows continuous monitoring of the benefit / risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system. "address: www.agenziafarmaco.gov.it/it/responsabili.
04.9 Overdose
A few cases of overdose with Copaxone (up to 80 mg glatiramer acetate) have been reported.These cases were not associated with adverse events other than those mentioned in section 4.8.
There is no clinical experience with doses above 80 mg of glatiramer acetate.
In clinical studies, daily doses of up to 30 mg of glatiramer acetate for up to 24 months were not associated with adverse events other than those mentioned in section 4.8.
In the event of an overdose, patients should be monitored and appropriate symptomatic and supportive therapy instituted.
05.0 PHARMACOLOGICAL PROPERTIES
05.1 Pharmacodynamic properties
Pharmacotherapeutic group: other cytokines and immunomodulators.
ATC code: L03AX13.
The mechanism or mechanisms by which glatiramer acetate acts in MS patients have not yet been fully elucidated. However, the product is believed to act by altering the immune processes currently believed to be responsible for the pathogenesis of MS. This hypothesis was supported by the results of studies conducted to deepen the knowledge concerning the pathogenesis of "experimental allergic encephalomyelitis (EAE), a" disease that, in various animal species, is induced by immunization against material containing myelin derived from the nervous system and often used as a model of MS in experimental animals Studies in animals and MS patients indicate that glatiramer acetate-specific suppressor T lymphocytes are induced and activated at the periphery following its administration.
Relapsing-remitting multiple sclerosis (RRMS):
A total of 269 patients were treated with Copaxone in three controlled studies. The first was a two-year study in 50 patients (Copaxone # = 25, placebo # = 25); who had been diagnosed with relapsing multiple sclerosis (MS) with phases of remission and at least two bouts of neurological dysfunction (exacerbations) in the previous two-year period by applying standard criteria then in force. The second study used the same inclusion criteria and included 251 patients treated for up to 35 months (Copaxone n = 125, placebo n = 126). The third study was a nine-month study including 239 patients (Copaxone n = 119 , placebo n. = 120) and where the inclusion criteria were similar to those of the first and second studies with the addition of a further criterion, namely that the patients had had at least one gadolinium-enhancing lesion evidenced by magnetic resonance imaging (MRI ).
In clinical trials performed in MS patients treated with Copaxone, a significant reduction in the number of relapses was observed compared to placebo.
In the largest controlled study that was performed, the relapse rate decreased by 32% from 1.98 in patients taking placebo to 1.34 in patients taking glatiramer acetate.
Exposure data for 103 patients treated with Copaxone for a total period of twelve years are available.
Copaxone also demonstrated beneficial effects over placebo on MRI parameters relevant to relapsing MS with remission.
However, Copaxone had no beneficial effect on the progression of disability in patients with relapsing MS with remission.
There is no evidence that Copaxone treatment has an effect on the duration or severity of relapses.
The usefulness of Copaxone in patients with primary or secondary progressive disease has not currently been demonstrated.
First clinical event indicative of multiple sclerosis:
A placebo-controlled study including 481 patients (Copaxone n = 243, placebo n = 238) was conducted in subjects with a single, well-defined, unifocal neurological manifestation and MRI features strongly suggestive of MS (at least two brain lesions , highlighted with T2-weighted MRI, greater than 6 mm in diameter). Any other pathology, with the exception of MS, which could better interpret the signs and symptoms observed in the patient had to be excluded. The placebo control period was followed by an open-label treatment phase: patients who had MS symptoms or who were asymptomatic for three years, whichever came first, were assigned treatment with the active substance open for a further period of two years, not exceeding a maximum total treatment duration of 5 years. Of the 243 patients initially randomized to Copaxone, 198 continued Copaxone treatment in the open-label phase. Of the 238 patients initially randomized to placebo, 211 transitioned to Copaxone treatment in the open-label phase.
During the placebo-controlled treatment period of up to three years, Copaxone delayed progression from the first clinical event to Clinically Defined Multiple Sclerosis (CDMS) according to the Poser criteria significantly both statistically and clinically corresponding to a reduction risk of 45% (hazard ratio (HR) = 0.55; 95% CI [0.40, 0.77], p = 0.0005). The percentage of patients who developed CDMS was 43% for the placebo group and 25% for the Copaxone group.
The favorable effect of Copaxone treatment over placebo was also demonstrated in two secondary endpoints on MRI parameters, namely the number of new T2-weighted lesions and the volume of T2 lesions.
Subgroup analyzes were performed post-hoc in patients with different baseline characteristics with the aim of identifying a population at high risk of developing a second attack. In subjects who had at least one T1-weighted gadolinium-enhanced lesion on MRI and 9 or more T2 lesions at baseline, switch to CDMS was evident in 50% of subjects treated with placebo compared to 28% of subjects treated with Copaxone for a period of 2.4 years. For subjects with 9 or more T2 lesions at baseline, switch to CDMS was evident in 45% of subjects treated with placebo compared with 26% of subjects treated with Copaxone over a 2.4 year period. However, the impact of early Copaxone treatment in relation to long-term disease evolution is also not known in these subgroups of high-risk patients, as the study design was primarily to assess the time between onset of the first clinical event and the second In any case, treatment should only be considered for patients considered to be at high risk.
The effect demonstrated in the placebo control phase was maintained over the long-term follow-up period of up to 5 years. The time to progression from the first clinical event to CDMS was prolonged with early treatment with Copaxone compared to treatment. delayed, showing a 41% risk reduction with early versus late treatment (hazard ratio = 0.59; 95% CI [0.44; 0.80], p = 0.0005). subjects who progressed to CDMS in the delayed treatment group was higher (49.6%) than that in the early treatment group (32.9%).
A consistent effect over time in favor of early versus late treatment was seen in the annualized number of lesions over the entire study period for the new gadolinium-enhancing T1 lesions (reduced by 54%; p
No relevant differences were observed between the early and delayed treatment group, neither in T1 hypointense lesion volume nor in brain atrophy over 5 years. However, the brain atrophy analysis, at the last observed value (adjusted for treatment exposure) showed a reduction in favor of early glatiramer acetate treatment (mean difference in percent brain volume change was 0.28%; p = 0.0209).
05.2 Pharmacokinetic properties
No pharmacokinetic studies have been performed in patients. The data obtained in vitro and limited data collected from healthy volunteers revealed that when glatiramer acetate is administered subcutaneously, the active substance is readily absorbed and that much of the dose is rapidly degraded into smaller fragments already in the subcutaneous tissue.
05.3 Preclinical safety data
Non-clinical data reveal no special hazard for humans based on studies of safety pharmacology, repeated dose toxicity, reproductive toxicity, genotoxicity or carcinogenicity, in addition to information included in other sections of the SmPC. Due to the lack of human pharmacokinetic data, it is not possible to establish the exposure margins between humans and animals.
Deposition of immune complexes in the renal glomeruli has been reported in a limited number of rats and monkeys treated for at least six months. In a two-year rat study, there was no indication of immune complex deposits in the renal glomeruli.
Following administration to sensitized animals (guinea pigs or rats), cases of anaphylaxis have been reported. It is not known whether these data are relevant to humans.
Injection site toxicity was common after repeated administration to animals.
06.0 PHARMACEUTICAL INFORMATION
06.1 Excipients
Mannitol
Water for injections
06.2 Incompatibility
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products.
06.3 Period of validity
2 years.
06.4 Special precautions for storage
Keep the pre-filled syringes in the outer carton to protect the medicine from light.
Store in a refrigerator (2 ° C - 8 ° C).
Do not freeze.
If it is not possible to store the pre-filled syringes in the refrigerator, they can be stored at room temperature (between 15 ° C and 25 ° C) once for up to 1 month.
After this month, if Copaxone 20 mg / ml pre-filled syringes have not been used and are still in their original packaging they should be placed in the refrigerator (2 ° C - 8 ° C).
06.5 Nature of the immediate packaging and contents of the package
A pre-filled syringe containing Copaxone solution for injection consists of a 1 ml type I colorless glass cylindrical syringe with an attached needle, a plastic plunger with a rubber stopper and a needle guard.
Copaxone is available in packs containing 7, 28 or 30 pre-filled syringes of 1 ml solution for injection and in a multipack containing 90 (3 packs of 30) pre-filled syringes of 1 ml solution for injection.
Not all pack sizes may be marketed.
06.6 Instructions for use and handling
The product is for single use only. Unused medicine and wastes derived from that medicine should be disposed of.
07.0 MARKETING AUTHORIZATION HOLDER
Teva Pharmaceuticals Ltd.
Ridings Point, Whistler Drive, Castleford
West Yorkshire, WF10 5HX (United Kingdom)
08.0 MARKETING AUTHORIZATION NUMBER
A.I.C. No. 035418021 "20 mg / ml solution for injection in pre-filled syringes" 28 syringes
A.I.C. No. 035418033 "20 mg / ml solution for injection in pre-filled syringes" 7 syringes
A.I.C. No. 035418045 "20 mg / ml solution for injection in pre-filled syringes" 30 syringes
A.I.C. No. 035418058 "20 mg / ml solution for injection in pre-filled syringes" 90 (3x30) syringes
09.0 DATE OF FIRST AUTHORIZATION OR RENEWAL OF THE AUTHORIZATION
Date of first authorization: 27 April 2005
Date of most recent renewal: March 23, 2009
10.0 DATE OF REVISION OF THE TEXT
July 2014