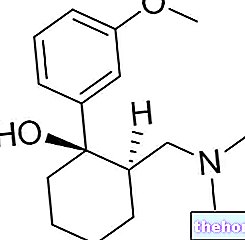
Active ingredients: Methyldopa
ALDOMET 250 mg film-coated tablets
ALDOMET 500 mg film-coated tablets
Why is Aldomet used? What is it for?
ALDOMET is an antihypertensive drug indicated in the treatment of moderate or severe arterial hypertension. It acts on the central nervous system with a sympathomimetic action.
Contraindications When Aldomet should not be used
Do not take ALDOMET
- if you are allergic (hypersensitive) to methyldopa or any of the other ingredients of ALDOMET.
- if you have or have suffered from liver disease, such as acute hepatitis and liver cirrhosis, and / or severe kidney disease with very high blood urea.
- if you take monoamine oxidase (MAO) inhibitor drugs.
- if you have high blood pressure caused by a pheochromocytoma.
Precautions for use What you need to know before taking Aldomet
Take special care with ALDOMET
- if anemia occurs: in this case appropriate laboratory tests should be performed to ascertain the possible presence of haemolysis. In the case of haemolytic anemia, treatment must be stopped. By stopping the administration of methyldopa or starting a corticosteroid treatment usually rapid remission of anemia is achieved. However, fatalities have rarely occurred.
- Before treatment with ALDOMET, your doctor should carefully evaluate the benefit / risk ratio, and you should consider starting treatment with ALDOMET only if other antihypertensive drugs have proved ineffective or are contraindicated in your case.
- In 10-20% of patients undergoing prolonged treatment with methyldopa a positive result was found in the direct Coombs test. A positive Coombs test is rarely found in the first six months of therapy with methyldopa; if this does not occur within 12 months it is unlikely that positivity will manifest itself with prolonged treatment. This phenomenon is dose-dependent, so it occurs with minimal frequency in patients who are treated with 1 g of methyldopa per day or with a lower dosage. After a few weeks, or a few months from the interruption of treatment, the Coombs test is again negative. In case of need for transfusions, being aware of a positive reaction to the Coombs test is useful for the evaluation of cross-compatibility tests. . Patients in whom a positive Coombs test is found during cross-tests may be incompatible in the minor cross-test. In this case it is advisable to perform the indirect Coombs test. If this is negative, the transfusion with the blood under examination - which on the other hand is compatible in the major cross-test - can certainly be carried out. However, if this is positive, it is up to the haematologist or transfusion expert to decide whether to transfuse with compatible blood in the major cross-test.
- A reversible reduction in the number of leukocytes (white blood cells) has rarely been observed, mainly involving the granulocytes. Upon discontinuation of treatment, the number of granulocytes readily returns to normal. Rare cases of agranulocytosis have been reported. In any case, interrupting the treatment resulted in a return to normal in the number of leukocytes.
- Fever has sometimes occurred in the first weeks of treatment with methyldopa; in some cases this was associated with eosinophilia or alterations in one or more liver function tests, such as: serum alkaline phosphatase, serum transaminases (SGOT, SGPT), bilirubin, cephalin-cholesterol flocculation, prothrombin time and bromosulfonphthalein retention. Jaundice may also occur with or without fever usually within the first two to three months of therapy. Signs of biliary retention have been observed in some patients. Rare cases of fatal hepatic necrosis have been reported. Liver biopsy, performed in several patients with hepatic dysfunction, showed a microscopic focal necrosis that could refer to drug hypersensitivity. If fever or abnormal liver function tests, or jaundice are found, methyldopa therapy should be discontinued. If the temperature or changes in liver function were related to methyldopa, they characteristically returned to normal after discontinuation of the drug. Patients should be closely monitored in order to detect any undesirable effects or unusual manifestations of drug idiosyncrasy.
- Hypertension may occasionally occur after dialysis in patients treated with ALDOMET because the drug is removed by this procedure.
- Involuntary choreo-athetotic movements have rarely been observed during methyldopa therapy in patients with severe bilateral cerebrovascular disease. If these movements occur, discontinue therapy.
- Methyldopa determines, in urine samples, fluorescence at the same wavelength as catecholamines: therefore, the determination of urinary catecholamines can give falsely elevated values, which can interfere with diagnostic tests for pheochromocytoma. The use of methyldopa cannot serve as a diagnostic tool for pheochromocytoma.
Interactions Which drugs or foods can modify the effect of Aldomet
Taking ALDOMET with other medicines
Tell your doctor if you are taking or have recently taken any other medicines, including medicines obtained without a prescription.
Lithium
During concomitant treatment with methyldopa and lithium, close monitoring for symptoms of lithium toxicity is required.
Other antihypertensive drugs
When methyldopa is used in combination with other antihypertensive drugs, potentiation of the antihypertensive action (hypotension) may occur.
Iron
Several studies show a decrease in the bioavailability of methyldopa when ingested with ferrous sulfate and ferrous gluconate. This can have a negative effect on blood pressure control in patients treated with methyldopa.
Monoamine oxidase (MAO) inhibitors
See "Do not take ALDOMET".
Other
During ALDOMET therapy you may need reduced doses of anesthetic. If hypotension occurs during anesthesia, it can usually be controlled by vasopressor drugs. The adrenergic receptors remain sensitive during treatment with methyldopa. In some patients undergoing prolonged treatment with methyldopa, a positive direct Coombs test was found; moreover other interference with laboratory tests has occurred (see "Take special care with ALDOMET").
Taking ALDOMET with food and drink
There is no interference of Aldomet with food and drink.
Warnings It is important to know that:
Pregnancy and breastfeeding
Ask your doctor for advice before taking any medicine. Since clinical experience and studies relating to the use of methyldopa in pregnancy have so far been limited, administration of the drug during pregnancy is not recommended. Methyldopa is excreted with breast milk. Therefore, the use of the drug in breastfeeding women requires that the benefits be weighed against the possible risks.
Driving and using machines
Do not use tools or machines if, during therapy, side effects occur that can negatively interfere such as impaired mental acuity and others.
Important information about some of the ingredients of ALDOMET
None.
Dosage and method of use How to use Aldomet: Dosage
Always take ALDOMET exactly as your doctor has told you. If in doubt, consult your doctor or pharmacist.
The usual starting dose, in the first 48 hours, is 250 mg two or three times a day. Thereafter, the daily dose can be adjusted, preferably at intervals of not less than two days, until an adequate response is achieved. Once the effective dose is reached, a gradual blood pressure response is seen in most patients as early as 12-24 hours. The recommended dosage of ALDOMET per day ranges from 500 mg up to 2 g in divided doses. Although some patients responded to higher doses, it was not useful to exceed the maximum daily dose of 3 g. The 500 mg tablets have been designed for the therapy of patients requiring two 250 mg tablets for each administered dose. This type of posology has not been studied for the initial therapy of previously untreated hypertensive patients.
Use with thiazide diuretics
If effective blood pressure control cannot be maintained with a daily dosage of 2 g of methyldopa, the combination with a thiazide diuretic is recommended. As thiazides have been shown to complement the efficacy of methyldopa, patients should be followed closely to detect changes in blood pressure. To prevent an excessive fall in blood pressure, the doctor may choose, as soon as a thiazide is associated, to reduce the dosage of ALDOMET by 50% or to continue, if preferred, with the entire dosage of ALDOMET by associating a thiazide with small Gradual dose increases in order to determine the potentiating effect. ALDOMET may be introduced in the antihypertensive therapy of patients treated with thiazides. The dosage of ALDOMET should initially be limited to no more than 375 mg per day during the first 48 hours and gradually increased. at intervals of not less than two days until an adequate response is obtained.
Transfer from other antihypertensive drugs
ALDOMET therapy can be initiated in patients already being treated with ganglionplegics or guanethidine. By gradually decreasing the dosage of these drugs and adding ALDOMET it is possible to obtain a gradual replacement of the therapy with an optimal pressure control.
ALDOMET therapy can be initiated in most patients already treated with other antihypertensive drugs (e.g. reserpine, other rauwolfia derivatives, hydralazine, monoamine oxidase inhibitors) by stopping the administration of these antihypertensive drugs. ALDOMET, subsequently administered to patients already treated with previous antihypertensive drugs, should be limited to an initial dose of no more than 375 mg per day and increased as needed at intervals of not less than two days.
General informations
Methyldopa is extensively eliminated by the kidney. Therefore, patients with impaired renal function may respond to drug doses lower than those required for patients with intact renal function. Cases of syncope, which occurred in older patients, have been linked to a greater sensitivity of patients with advanced arteriosclerotic vasculopathy: this can be avoided by reducing the dosage of ALDOMET. A sedative effect occurs in many patients for two to three days after initiation of ALDOMET therapy or when the dose is increased. The sedative effect usually disappears when an effective maintenance dose is achieved. When the dosage is increased it is advisable to start by increasing the evening dose; in this way the sedative effect is minimized without the postural hypotension in the morning being accentuated.Sometimes, both in the initial and in the advanced stages of treatment, addiction can occur; however, this is more likely to occur between the second and third month of therapy. Increasing the dose of ALDOMET or adopting an associated therapy with thiazides frequently restores effective control of blood pressure. Since methyldopa has a relatively short duration of action, discontinuation of treatment is followed by a gradual return to previous blood pressure levels usually within 48 hours. This is not complicated by a pressure rebound.
Use in children
ALDOMET is not indicated for children.
Overdose What to do if you have taken too much Aldomet
If you take more ALDOMET than you should
Acute overdose can produce acute hypotension and other reactions attributable to cerebral and gastrointestinal dysfunction (excessive sedation, weakness, bradycardia, dizziness, light-headedness, constipation, distension, flatulence, diarrhea, nausea, vomiting). There is no specific antidote. In the event of an overdose, symptomatic and supportive measures should be used. If ingestion is recent, gastric lavage or vomiting may reduce absorption; when more time has passed since ingestion, to promote urinary excretion, drips can be helpful. , urinary function and brain activity. The administration of sympathomimetic drugs may be indicated. Methyldopa is dialyzable.
If you forget to take ALDOMET
Do not take a double dose to make up for a forgotten tablet.
If you stop taking ALDOMET
If you have any further questions on the use of ALDOMET, ask your doctor or pharmacist.
Side Effects What are the side effects of Aldomet
Like all medicines, ALDOMET can cause side effects, although not everybody gets them.
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
At the start of treatment or when the dosage is increased, a sedative effect, usually transient, may be found. In the initial phase of treatment, transient effects such as headache, asthenia and weakness may be seen.
The following undesirable reactions have been reported during treatment with ALDOMET:
Nervous system disorders: sedation (usually transient), headache, asthenia or weakness, paraesthesia, parkinsonism, Bell's palsy, involuntary choreoathetotic movements, mental disorders including nightmares, impaired mental acuity and mild reversible psychosis or depression. Dizziness, light-headedness, and symptoms of cerebrovascular insufficiency (may be due to low blood pressure).
Cardiac and vascular disorders: Bradycardia, prolonged hypersensitivity of the carotid sinus, aggravation of an "angina pectoris. Orthostatic hypotension (decrease daily dosage). Edema (and weight gain) usually reversible by administration of a diuretic; if edema progresses or signs of heart failure appear, drug administration should be suspended.
Gastrointestinal disorders: nausea, vomiting, bloating, constipation, flatulence, diarrhea, colitis, mild dry mouth, glossodynia or glossophyteia, pancreatitis, sialadenitis.
Hepatobiliary disorders: liver disorders including hepatitis, jaundice, abnormal liver function tests.
Blood and lymphatic system disorders: positive Coombs test, haemolytic anemia, bone marrow depression, leukopenia, granulocytopenia, thrombocytopenia, eosinophilia. Positive tests for antinuclear antibodies, LE cells and rheumatoid factor.
Allergic disorders: drug fever and lupus-like syndrome, myocarditis, pericarditis.
Skin and subcutaneous tissue disorders: rash as in eczema and lichenoid eruptions, toxic epidermal necrolysis.
Endocrine disorders: breast enlargement, gynaecomastia, lactation, hyperprolactinaemia, amenorrhea, impotence, decreased libido.
Musculoskeletal and connective tissue disorders: mild arthralgia, with or without joint swelling, myalgia.
Respiratory, thoracic and mediastinal disorders: sense of nasal occlusion.
Diagnostic tests: increased azotemia.
Expiry and Retention
Store in the original packaging. Do not use ALDOMET after the expiry date which is stated on the carton after EXP. The expiry date refers to the last day of the month.
Keep this medicine out of the reach and sight of children.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. This will help protect the environment.
Composition and pharmaceutical form
What ALDOMET contains
- The active ingredient is: methyldopa
- The excipients are:
Excipients of the tablet: sodium calcium edetate; ethylcellulose; guar gum; pulverized cellulose; colloidal hydrated silica; magnesium stearate; carnauba wax.
Excipients of the film: Opadry 03H38061 yellow (citric acid anhydrous, hypromellose 2910, E172 red iron oxide, propylene glycol, E 104 quinoline yellow, talc, E171 titanium dioxide). -
Description of how ALDOMET looks and contents of the pack
ALDOMET 250: lithographed cardboard box containing 30 film-coated tablets of 250 mg in blister packs.
ALDOMET 500: lithographed cardboard box containing 30 film-coated tablets of 500 mg in blister.
Source Package Leaflet: AIFA (Italian Medicines Agency). Content published in January 2016. The information present may not be up-to-date.
To have access to the most up-to-date version, it is advisable to access the AIFA (Italian Medicines Agency) website. Disclaimer and useful information.
01.0 NAME OF THE MEDICINAL PRODUCT
ALDOMET
02.0 QUALITATIVE AND QUANTITATIVE COMPOSITION
Each 250 mg film-coated tablet contains: 250 mg of methyldopa.
Each 500 mg film-coated tablet contains: 500 mg of methyldopa.
For excipients, see 6.1
03.0 PHARMACEUTICAL FORM
Film-coated tablets
04.0 CLINICAL INFORMATION
04.1 Therapeutic indications
ALDOMET is indicated for the treatment of moderate or severe hypertension.
04.2 Posology and method of administration
ALDOMET is available in tablets of 250 and 500 mg of methyldopa, for oral use.
Therapy is usually started by administering 250 mg two or three times a day in the first 48 hours.
Thereafter, the daily dose can be adjusted, preferably at intervals of not less than two days, until an adequate response is achieved. Once the effective dose is reached, a gradual blood pressure response is seen in most patients as early as 12-24 hours.
The recommended dosage of ALDOMET per day ranges from 500 mg up to 2 g in divided doses. Although some patients responded to higher doses, it was not useful to exceed the maximum daily dose of 3 g.
The 500 mg tablets have been designed for the therapy of patients requiring two 250 mg tablets for each administered dose. This type of posology has not been studied for the initial therapy of previously untreated hypertensive patients.
Use with thiazides
If effective control of blood pressure cannot be maintained with a daily dosage of 2 g of methyldopa, the combination with a thiazide diuretic is recommended.
Since thiazides have been shown to complement the efficacy of methyldopa, patients should be followed closely in order to detect changes in blood pressure. To prevent an excessive fall in blood pressure, the doctor may choose, as soon as a thiazide is combined, to reduce by 50% the dosage of ALDOMET or to continue, if preferred, with the entire dosage of ALDOMET associating a thiazide with small gradual increases of the dosage in order to determine the potentiating effect. ALDOMET can be introduced in the antihypertensive therapy of patients treated with thiazides. The dosage of Aldomet should be initially limited to no more than 375 mg per day during the first 48 hours and gradually increased at intervals of not less than two days until an adequate response is achieved.
Transfer from other antihypertensive drugs.
ALDOMET therapy can be initiated in most patients already treated with other antihypertensive drugs by stopping the administration of these antihypertensive drugs gradually if required (consult the technical data sheet before discontinuing these drugs).
ALDOMET, subsequently administered to patients already treated with previous antihypertensive drugs, should be limited to an initial dose of no more than 375 mg per day and increased as needed at intervals of not less than two days.
General informations
Methyldopa is extensively eliminated by the kidney. Therefore, patients with impaired renal function may respond to drug doses lower than those required for patients with intact renal function. Cases of syncope, which occurred in older patients, have been linked to a greater sensitivity of patients with advanced arteriosclerotic vasculopathy: this can be avoided by reducing the dosage of ALDOMET.
A sedative effect occurs in many patients for two to three days after initiation of ALDOMET therapy or when the dose is increased.
When the dosage is increased it is advisable to start by increasing the evening dose; in this way the sedative effect is minimized without the postural hypotension in the morning being accentuated.
Sometimes, both in the initial phase and in the advanced phase of treatment, addiction can occur; however, this is more likely to occur between the second and third month of therapy. Increasing the dose of ALDOMET or adopting an associated therapy with thiazides frequently restores effective control of blood pressure. As methyldopa has a relatively short duration of action, discontinuation of treatment is followed by a gradual return to previous blood pressure levels usually within 48 hours.
This is not complicated by a pressure rebound.
04.3 Contraindications
Active liver disease, such as acute hepatitis and active cirrhosis; severe nephropathies with very high azotemia values, hypersensitivity (including liver disorders associated with previous methyldopa therapy).
Methyldopa should be used with caution in patients with a history of liver disease or liver dysfunction (see Special warnings and precautions for use).
Methyldopa is not recommended for the treatment of patients with pheochromocytoma.
04.4 Special warnings and appropriate precautions for use
Rare cases of acquired haemolytic anemia have occurred during treatment with methyldopa. In consideration of this risk, the use of the product will be limited, after careful evaluation of the risk-benefit ratio by the doctor, to cases in which other antihypertensive drugs of habitual use have proved ineffective or contraindicated.
In the presence of clinical symptoms indicating the possibility of anemia, hemoglobin and / or hematocrit should be determined.
In the case of anemia, appropriate laboratory tests should be performed to ascertain the possible presence of haemolysis. In the case of established haemolytic anemia, the treatment must be interrupted. rapid remission of anemia. However, fatalities have rarely occurred.
In some patients undergoing prolonged treatment with methyldopa, a positive direct Coombs test was found. It has been reported by several investigators that the incidence of Coombs test positivity ranged from ten to twenty percent. Coombs test positivity is rarely found in the first six months of methyldopa therapy; if this does not occur within 12 months it is unlikely that positivity will manifest itself by prolonging the treatment.This phenomenon is dose-dependent, so it occurs with minimal frequency in patients who are treated with 1 g of methyldopa per day or with a lower dosage.
After a few weeks, or a few months from the interruption of treatment, the Coombs test is again negative. In case of need for transfusions, being aware of a positive reaction to the Coombs test is useful for the evaluation of cross-tests of compatibility. Patients in whom a positive Coombs test is found during cross-tests may be incompatible in the minor cross-test. In this case it is advisable to perform the indirect Coombs test. If this is negative, the transfusion with the blood under examination - which on the other hand is compatible in the major cross-test - can certainly be carried out. However, if this is positive, it is up to the haematologist or transfusion expert to decide whether to transfuse with compatible blood in the major cross-test. A reversible reduction in the number of leukocytes has rarely been observed, with the main involvement of the granulocytes.
On discontinuation of treatment the number of granulocytes promptly returned to normal. There have been rare cases of reversible thrombocytopenia.
Fever has occasionally occurred in the first 3 weeks of treatment with methyldopa; in some cases this was associated with eosinophilia or alterations in one or more liver function tests, such as: serum alkaline phosphatase, serum transaminases (SGOT, SGPT), bilirubin, cephalin-cholesterol flocculation, prothrombin time and bromosulfonphthalein retention.
Jaundice may also occur with or without fever usually within the first two to three months of therapy. Signs of colostasis have been observed in some patients.
Rare cases of fatal hepatic necrosis have been reported. Liver biopsy, performed in several patients with hepatic dysfunction, showed a microscopic focal necrosis that could refer to drug hypersensitivity.
A determination of liver function and white count with formula should be done at intervals during the first 6-12 weeks of therapy, or if unexplained fever develops.
If fever or abnormal liver function tests, or jaundice are found, methyldopa therapy should be discontinued. If the temperature or changes in liver function were related to methyldopa, they characteristically returned to normal after discontinuation of the drug.
Methyldopa should not be re-administered in these patients. Methyldopa should be used with caution in patients with a history of previous liver disease or dysfunction.
Patients should be closely monitored in order to detect any side effects or unusual manifestations of drug idiosyncrasy.
Hypertension may occur after dialysis in patients treated with ALDOMET because the drug is removed by this procedure.
Involuntary choreo-athetotic movements have rarely been observed during methyldopa therapy in patients with severe bilateral cerebrovascular disease. If these movements occur, discontinue therapy.
Methyldopa can interfere with the determination of uric acid carried out by the phosphotungstate method, of serum creatinine by the alkaline picrate method and of SGOT by the colorimetric method. Regarding the SGOT, no interference with the spectrophotometric methods has been reported. .
Methyldopa determines, in urine samples, fluorescence at the same wavelength as catecholamines: therefore, the determination of urinary catecholamines can give falsely elevated values, which can interfere with diagnostic tests for pheochromocytoma.
It is important to recognize this phenomenon before a patient with a possible pheochromocytoma undergoes surgery. Methyldopa does not interfere with the determinations of vanylmandelic acid (VMA) carried out by those methods that convert the VMA into vanillin.
Rarely, when the urine is exposed to air after urination, it can darken due to the breakdown of methyldopa or its metabolites.
04.5 Interactions with other medicinal products and other forms of interaction
When methyldopa is used in combination with other antihypertensive drugs, potentiation of the antihypertensive action may occur.
Patients on ALDOMET therapy may need reduced doses of anesthetic. If hypotension occurs during anesthesia, it can usually be controlled by vasopressor drugs.
The adrenergic receptors remain sensitive during treatment with methyldopa.
04.6 Pregnancy and lactation
Pregnancy
ALDOMET has been used in the treatment of hypertension in pregnancy, under close medical supervision and obstetric supervision. There has been no evidence that ALDOMET causes fetal abnormalities or damages the newborn.
Feeding time
Methyldopa crosses the placenta and appears in umbilical cord blood and breast milk.
Although no certain teratogenic effects have been reported, the possibility of fetal harm cannot be excluded and the use of the drug in pregnant or fertile or breastfeeding women requires that the benefits be weighed against the possible risks.
04.7 Effects on ability to drive and use machines
If, during therapy, side effects occur that can negatively interfere such as impaired mental acuity and others, avoid driving or operating machinery or otherwise carrying out activities that require prompt vigilance.
04.8 Undesirable effects
Significant side effects of ALDOMET were infrequent.
At the start of treatment or when the dosage is increased, a usually transient sedative effect may be found.
In the initial phase of treatment, transient effects such as headache, asthenia and weakness may be seen.
The following undesirable reactions have been reported during treatment with ALDOMET:
Central Nervous System: sedation (usually transient), headache, asthenia or weakness, paraesthesia, parkinsonism, Bell's palsy, involuntary choreoathetotic movements, mental disorders including nightmares, impaired mental acuity and mild reversible psychosis or depression. Vertigo, light-headedness, and symptoms of cerebrovascular insufficiency (may be due to low blood pressure).
Cardiovascular: Bradycardia, prolonged hypersensitivity of the carotid sinus, aggravation of angina pectoris. Orthostatic hypotension (decrease daily dosage). Edema (and weight gain) usually reversible by administration of a diuretic. If edema progresses or signs of heart failure, drug administration should be suspended.
Gastrointestinal: nausea, vomiting, bloating, constipation, flatulence, diarrhea, colitis, mild dry mouth, glossodynia or glossophyteia, pancreatitis, sialadenitis.
Hepatic: liver disorders including hepatitis, jaundice, abnormal liver function tests.
Haematological: positive Coombs test, haemolytic anemia, bone marrow depression, leukopenia, granulocytopenia, thrombocytopenia. Positive tests for antinuclear antibodies, LE cells and rheumatoid factor.
Allergic: drug-related fever and lupus-like syndrome, myocarditis, pericarditis.
Dermatological: rash as in eczema and lichenoid eruptions, toxic epidermal necrolysis.
Others: sense of nasal occlusion, increased azotemia, breast enlargement, gynecomastia, lactation, hyperprolactinemia, amenorrhea, impotence, decreased libido, mild arthralgia, with or without joint swelling, myalgia.
04.9 Overdose
Acute overdose can produce acute hypotension and other reactions attributable to cerebral and gastrointestinal dysfunction (excessive sedation, weakness, bradycardia, dizziness, light-headedness, constipation, distension, flatulence, diarrhea, nausea, vomiting).
There is no specific antidote. In the event of an overdose, symptomatic and supportive measures should be used. If ingestion is recent, gastric lavage or vomiting may reduce absorption; when more time has passed since ingestion, to promote urinary excretion, drips can be helpful. , urinary function and brain activity. The administration of sympathomimetic drugs may be indicated. Methyldopa is dialyzable.
05.0 PHARMACOLOGICAL PROPERTIES
05.1 Pharmacodynamic properties
ALDOMET is an antihypertensive which reduces blood pressure in both the reclining and orthostatic positions.
The antihypertensive effect is probably due to its metabolism to alpha-methylnoradrenaline which lowers blood pressure by stimulating central alpha-adrenergic inhibitory receptors by acting as a false neurotransmitter and / or by reducing plasma renin activity.
Symptomatic postural hypotension, exertional hypotension, and diurnal changes in blood pressure occur rarely with ALDOMET.
It has no direct effect on cardiac function and generally does not reduce glomerular filtration rate, renal blood flow or filtration fraction.
Cardiac output generally remains unchanged, with no increase in heart rate occurring; in some patients the heart rate decreased. Due to its relative lack of adverse effects on renal function, methyldopa can be used advantageously to control arterial hypertension, even in the presence of renal insufficiency. ALDOMET can help stop or delay the progression of renal insufficiency and the damage due to it. the prolonged increase in blood pressure.
05.2 "Pharmacokinetic properties
Absorption of methyldopa shows large individual variations. In two studies, its bioavailability was in the range of 8% to 62%.
05.3 Preclinical safety data
The pharmacological and toxicological laboratory tests relating to methyldopa are very interesting, given its close structural similarity with amino acids, natural precursors of the adrenergic mediation of the autonomic system. For example, the acute intravenous LD50 is 1900 mg / kg in mice: that is, the drug is less toxic than dopa. Acute toxicity, for oral administration, ranges from 5300 mg / kg to over 15000 mg / kg depending on the vehicle. Studies on subacute oral toxicity in dogs performed with doses up to 2000 mg / kg per day for four weeks, did not reveal histopathological changes related to the administration of methyldopa, although inanition was observed at maximum doses.
Chronic oral toxicity studies conducted over long periods at doses up to 1800 mg / kg / day in rats and 1350 mg / kg / day in dogs and 1000 mg / kg / day in monkeys did not reveal histopathological and chemical changes of clinical importance. .
Reproduction studies conducted with methyldopa in 3 successive generations of mice and in two successive generations of rats, as well as from teratogenic studies in rabbits, did not reveal any side effects.
06.0 PHARMACEUTICAL INFORMATION
06.1 Excipients
Each film-coated tablet contains the following excipients: anhydrous citric acid; sodium calcium edetate; ethylcellulose; guar gum; pulverized cellulose; colloidal hydrated silica; magnesium stearate; hypromellose; propylene glycol; titanium dioxide; talc; red iron oxide; E 104 quinoline yellow on hydrated aluminum; carnauba wax.
06.2 Incompatibility
Incompatibilities with other medicines are unknown.
06.3 Period of validity
36 months
06.4 Special precautions for storage
There are no special storage precautions.
06.5 Nature of the immediate packaging and contents of the package
The film-coated tablets are contained in a PVC and aluminum blister
30 tablets 250 mg
30 tablet 500 mg
06.6 Instructions for use and handling
No special instructions.
07.0 MARKETING AUTHORIZATION HOLDER
Merck Sharp & Dohme Ltd. - Hoddesdon - England
Subsidiary of MERCK & Co. Inc., Whitehouse Station, N.J., U.S.A.
Exclusive representative for Italy
MERCK SHARP & DOHME (Italy) S.p.A.
Via G. Fabbroni, 6 - 00191 Rome
08.0 MARKETING AUTHORIZATION NUMBER
30 tab 250 mg 019954039
30 tablet 500 mg 019954015
09.0 DATE OF FIRST AUTHORIZATION OR RENEWAL OF THE AUTHORIZATION
June 2000
10.0 DATE OF REVISION OF THE TEXT
July 2000










.jpg)