Watch the video
- Watch the video on youtube

According to the triggering causes, it is possible to distinguish two different types of carnitine deficiency: primary and secondary.
To better understand the causes and consequences of carnitine deficiency, it is certainly useful to know this molecule and the biological functions it performs.
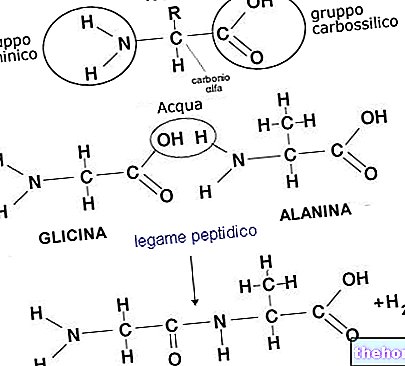
produced by the body - mainly by the liver and kidneys - starting from two essential amino acids, lysine and methionine. At the cellular level, carnitine plays the role of transporter of long-chain fatty acids inside the mitochondrion, where they will be subjected to beta -oxidation in order to produce ATP molecules, an indispensable source of energy for carrying out all cellular activities. Carnitine therefore plays an essential role in energy production.
To carry out its function, however, carnitine must be able to enter the cell, bind to the fatty acids found in the cytoplasm, transport them inside the mitochondrion and release them, and then exit the mitochondrial matrix and start a new cycle. , for all this to happen, both the presence of a suitable concentration of carnitine and the presence and functioning of a system of proteins and transporters localized on the cellular and mitochondrial membrane are necessary. The set of reactions that allow carnitine to carry out its task, involving also the proteins and transporters mentioned above, is called the "carnitine system".
Therefore, for the production of energy to take place correctly, it is essential that this whole system is fully functional.
Alterations and anomalies of any kind in any of the steps or factors that make up the carnitine system, can unbalance the production of energy, therefore the cellular metabolism and the functions of the different tissues of the organism, leading to the appearance of diseases often serious and fatal.
which occur on genes coding for proteins involved in the carnitine system. Depending on the gene affected by the mutation, it is possible to distinguish several types of primary carnitine deficiency. However, without going into in-depth descriptions, we will limit ourselves to saying that the proteins involved can be:
- Carnitine-palmitoyl-transferase type I (CPT-I): it is localized on the external mitochondrial membrane, catalyzes the transfer of acyl groups from acyl-CoA to carnitine, producing acylcarnitines.
- Carnitine-palmitoyl-transferase type II (CPT-II): it is localized on the inner mitochondrial membrane, catalyzes the reverse reaction of that catalyzed by CPT-I, converting the acylcarnitines into acyl-CoA, then conveyed to beta-oxidation.
- Carnitine-acylcarnitine-translocase (CT or CACT): it is localized on the inner mitochondrial membrane; exchanges cytoplasmic acylcarnitine with mitochondrial free carnitine (antiport mode), exports acetylcarnitine from the mitochondrion to the cytosol (uniport mode), allows the flow of carnitine and short-chain acylcarnitine from the mitochondrion to the cytosol and vice versa, in order to maintain a correct free carnitine / acylcarnitine ratio.
Mutations in these proteins lead to a drastic decrease in intracellular carnitine levels. As a result, long-chain fatty acids cannot be transferred into the mitochondrial matrix and are no longer available for beta-oxidation and energy production.