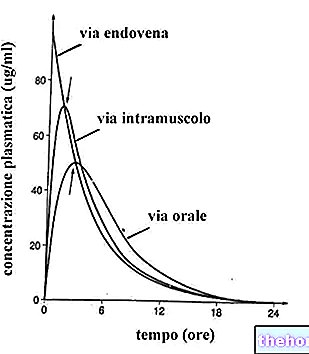
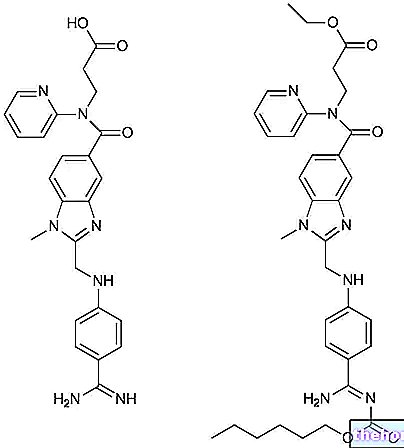
PARIET ® is a drug based on Rabeprazole sodium.
THERAPEUTIC GROUP: Antireflux - Antiulcer-Inhibitors of the proton pump

Indications PARIET ® Rabeprazole
PARIET ® is useful in the treatment of all those affections of the gastro-iintestinal tract, associated with increased gastric acidity, such as duodenal ulcers, gastric ulcer, gastroesophageal reflux disease, esophagitis and Zollinger-Ellison syndrome.
In combination with antibiotics PARIET ® can also be used in the eradication therapy of Helicobacter Pylori.
Mechanism of action PARIET ® Rabeprazole
The rabeprazole contained in PARIET ® belongs to the benzimidazole derivatives and to the broader category of proton pump inhibitors.
Its therapeutic efficacy, in fact, is due to the ability, once absorbed via the intestine and distributed through the bloodstream bound to plasma proteins, to activate in an acid environment such as the gastric canaliculi and selectively inhibit the H + / K + ATPase pump, involved in secretion. of hydrogen ions in the intragastric lumen, and expressed on the surface of the parietal cells.
More precisely, the inhibitory action is observed after a few hours, to be optimized around the third day, with a reduction of the intragastric acid content of about 80%.
Following hepatic metabolism, supported by highly polymorphic enzymes such as cytochrome p450 (CYP2C19 and CYP3A4), the metabolites of rabeprazole are eliminated predominantly in the urine as inactive compounds.
Studies carried out and clinical efficacy
1. EFFECTIVENESS AND SAFETY OF LONG-TERM RABEPRAZOLE THERAPY
Intern Med. 2011; 50: 179-188. Epub 2011 Feb 1.
Safety and Efficacy of Long-Term Maintenance Therapy with Oral Dose of Rabeprazole 10 mg Once Daily in Japanese Patients with Reflux Esophagitis.
Fujimoto K, Hongo M; The Maintenance Study Group.
Rabeprazole is also very well suited to long-term treatment of gastroesophageal reflux. This study, conducted on Japanese patients who took this drug for about 2 years, demonstrated an important regression of symptoms, without the appearance of significant gastric lesions, despite the appearance in a few cases of polyps or cysts without tumor markers. Although this effect has not yet been demonstrated, it could be associated with the increased secretion of gastrin, induced by the drug.
2. RABEPRAZOLE AND GASTRIC ULCERS
J Gastroenterol. 2011 Feb 26.
A randomized controlled trial of rebamipide plus rabeprazole for the healing of artificial ulcers after endoscopic submucosal dissection.
Fujiwara S, Morita Y, Toyonaga T, Kawakami F, Itoh T, Yoshida M, Kutsumi H, Azuma T.
Surgical treatment of gastric carcinoma removal evidently determines the appearance of lesions and ulcers of the gastric mucosa. The concomitant therapy between rabeprazole and rebapimide has been shown to be particularly effective in reducing symptoms, significantly facilitating the healing of the ulcer induced by the intervention.
3. COMPARISON OF THE PROTON PUMP INHIBITORS
Helicobacter. 2010 Oct; 15: 449-59.
Aliment Pharmacol Ther. 2011 Apr; 33: 845-54. doi: 10.1111 / j.1365-2036.2011.04580.x.
An open-label, parallel, multiple-dose study comparing the pharmacokinetics and gastric acid suppression of rabeprazole extended-release with esomeprazole 40 mg and rabeprazole delayed-release 20 mg in healthy volunteers.
Morelli G, Chen H, Rossiter G, Rege B, Lu Y.
This interesting study has shown how controlled release rabeprazole can guarantee a longer duration of action of the suppression of acid secretion, even 24 hours longer than that induced by esomeprazole or standard formulations of the same active ingredient.
Method of use and dosage
PARIET ® gastro-resistant tablets of 10 - 20 mg of rabeprazole:
a daily dose of 20 mg of raberazole, taken in a single morning administration, was found to be particularly effective in reducing the symptoms associated with gastroesophageal reflux and in healing duodenal and gastric ulcers in just 4-8 weeks of treatment.
Despite the aforesaid dosage, represents the most clinically used, the assumption of rabeprazole can be subject to important variations in terms of dosage according to the physio-pathological conditions of the patient and his therapeutic objectives.
Consequently, the intake of PARIET ® should be regulated and supervised by a competent physician.
PARIET ® Rabeprazole warnings
The intake of PARIET ® in patients with reduced renal and hepatic function should be done under strict medical supervision, given the presence of studies showing how a reduced hepatic metabolism can lead to an increase in even significant blood concentrations of rabeprazole.
Before starting the intake of rabeprazole, as well as for all inhibitors of the proton acid pump, it would be important to exclude malignant forms of gastrointestinal diseases, the symptoms of which could be masked by the therapeutic effects of the drug during its intake.
Although the administration of rabeprazole, especially when continued over time, is responsible for a significant increase in blood levels of gastrin, no incidence of pathologies or histological lesions of the gastric mucosa has been observed.
The presence of side effects such as headache, dizziness and drowsiness could impair the normal ability to drive and use machines.
PREGNANCY AND BREASTFEEDING
The intake PARIET ® is contraindicated during pregnancy and during the subsequent lactation period, given the absence of clinical trials or experimental models capable of certifying the absence of toxicity for the health of the fetus and the infant, following intake of rabeprazole during pregnancy or breastfeeding.
Interactions
Although rabeprazole is metabolised by cytochrome p450 enzymes, which are involved in the metabolism of numerous active substances, there was no interference with the normal metabolic activities of these enzymes.
Therefore, in most cases, the main pharmacokinetic variations induced by PARIET ® are essentially due to the significant reduction of the acid content of the stomach, which can reduce the absorption of different drugs such as antifungals.
Contraindications PARIET ® Rabeprazole
PARIET ® is contraindicated in pregnancy and lactation and in patients with known hypersensitivity to rabeprazole or other active substances belonging to the category of proton pump inhibitors.
Undesirable Effects - Side Effects
The rabeprazole contained in PARIET ® has proved to be quite well tolerated despite the presence of side effects such as cough, pharyngitis, rhinitis, diarrhea, nausea, vomiting, flatulence, constipation, abdominal pain, insomnia, dizziness and clinically insignificant headache.
More serious adverse reactions such as nephritis, gastritis, cutaneous, visual and haematological disorders, have been observed very rarely and especially in particular categories of patients at risk.
Note
PARIET ® sold only under medical prescription.
The information on PARIET ® Rabeprazole published on this page may be out of date or incomplete. For a correct use of this information, see the Disclaimer and useful information page.