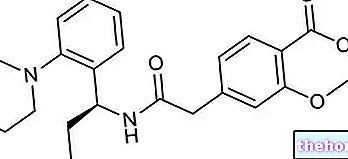
Active ingredients: Otilonium bromide
SPASMOMEN 40 mg coated tablets
Indications Why is Spasmomen used? What is it for?
Pharmacotherapeutic group
Synthetic anticholinergics, quaternary ammonium compounds.
Therapeutic indications
Irritable colon and spastic-painful manifestations of the distal enteric tract.
Contraindications When Spasmomen should not be used
Hypersensitivity to the active substance or to any of the excipients.
Precautions for use What you need to know before taking Spasmomen
To be used with caution in subjects with glaucoma, prostatic hypertrophy and pyloric stenosis.
Interactions Which drugs or foods can change the effect of Spasmomen
Tell your doctor or pharmacist if you have recently taken any other medicines, even those without a prescription.
Warnings It is important to know that:
Pregnancy and breastfeeding
Ask your doctor or pharmacist for advice before taking any medicine.
Although no embryotoxic, teratogenic or mutagenic effects have been reported in animals, as for all drugs its use in pregnancy and during lactation must be limited to cases of recognized necessity and under the direct supervision of the physician.
Effects on ability to drive and use machines
SPASMOMEN does not affect the ability to drive or use machines.In case of ascertained intolerance to sugars, contact your doctor before taking the medicine.
Dose, Method and Time of Administration How to use Spasmomen: Posology
1 coated tablet 2-3 times a day, according to the doctor's judgment.
Duration of treatment According to medical prescription.
Overdose What to do if you have taken too much Spasmomen
In animals, otilonium bromide has been shown to be practically free of toxicity. Consequently, also in the man should not arise particular problems due to overdose.
In this specific case, a possible symptomatic and supportive therapy is recommended.
In case of accidental ingestion / intake of an excessive dose of SPASMOMEN, notify your doctor immediately or go to the nearest hospital.
IF YOU HAVE ANY DOUBT ABOUT THE "USE OF SPASMOMEN, CONTACT YOUR DOCTOR OR PHARMACIST
Side Effects What are the side effects of Spasmomen
Like all medicines, SPASMOMEN can cause side effects, although not everybody gets them.
The following undesirable effects have been reported since marketing: Urticaria
Compliance with the instructions contained in the package leaflet reduces the risk of undesirable effects.
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
Expiry and Retention
Expiry: See the expiry date printed on the package.
The expiry date refers to the product in intact packaging, correctly stored.
Warning: do not use the medicine after the expiry date shown on the package.
Medicines should not be disposed of via wastewater or household waste. Ask your doctor or pharmacist for advice on throwing away medicines you no longer use. This will help protect the environment.
KEEP OUT OF THE SIGHT AND REACH OF CHILDREN
It is important to always have the information on the medicine available, therefore keep both the box and the package leaflet.
The expiry date indicated refers to the last day of the month.
Do not use this medicine if you notice visible signs of deterioration.
Composition and pharmaceutical form
Composition
Each coated tablet contains:
Active ingredient: otilonium bromide 40 mg.
Excipients - Core: lactose, starch, sodium starch glycolate, magnesium stearate.
Coating: hypromellose, titanium dioxide, macrogol, talc.
Pharmaceutical form and content
40 mg coated tablets, for oral use.
30 tablets
Source Package Leaflet: AIFA (Italian Medicines Agency). Content published in January 2016. The information present may not be up-to-date.
To have access to the most up-to-date version, it is advisable to access the AIFA (Italian Medicines Agency) website. Disclaimer and useful information.
01.0 NAME OF THE MEDICINAL PRODUCT
SPASMOMEN - Tablets
02.0 QUALITATIVE AND QUANTITATIVE COMPOSITION
Each coated tablet contains:
active principle: otylonium bromide 40 mg.
For excipients, see section 6.1
03.0 PHARMACEUTICAL FORM
Coated tablets.
04.0 CLINICAL INFORMATION
04.1 Therapeutic indications
Irritable colon and spastic-painful manifestations of the distal enteric tract.
04.2 Posology and method of administration
1 coated tablet 2-3 times a day, according to the doctor's judgment.
04.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients.
04.4 Special warnings and appropriate precautions for use
To be used with caution in subjects with glaucoma, prostatic hypertrophy, pyloric stenosis.
The medicinal product contains lactose, therefore it is not suitable for subjects with lactase deficiency, galactosemia or glucose / galactose malabsorption syndrome.
04.5 Interactions with other medicinal products and other forms of interaction
None.
04.6 Pregnancy and lactation
Although no embryotoxic, teratogenic or mutagenic effects have been reported in animals, as for all drugs, its use in pregnant women and during lactation should be limited in cases of recognized necessity and under direct medical supervision.
04.7 Effects on ability to drive and use machines
No effects on the ability to drive and use machines are known to date.
04.8 Undesirable effects
At therapeutic doses the product does not give rise to side effects and in particular does not cause atropine-like effects.
04.9 Overdose
In animals, otilonium bromide has been shown to be practically free of toxicity. Consequently, also in man should not arise particular problems due to overdose. In this specific case, a possible symptomatic and supportive therapy is recommended.
05.0 PHARMACOLOGICAL PROPERTIES
05.1 Pharmacodynamic properties
Pharmacotherapeutic group: Synthetic anticholinergics, quaternary ammonium compounds
ATC code: A03AB06
Otilonium bromide has an intense spasmolytic action on the smooth muscles of the digestive system.
05.2 Pharmacokinetic properties
Experimental data have shown that after oral administration, absorption is very poor and most of the absorbed portion is eliminated via the biliary route.
05.3 Preclinical safety data
Acute toxicity: per os no mortality up to 1500 mg / kg in rats and up to 1000 mg / kg in dogs.
Chronic toxicity: in experimental animals the oral administration of otilonium bromide at a dose of 80 mg / kg administered for 180 days did not cause any modification of the haematochemical and histological tests.
Teratology: no embryotoxic or teratogenic effects in rats and rabbits even for doses of 60 mg / kg.
Mutagenesis: no mutagenic effect in the numerous tests carried out.
06.0 PHARMACEUTICAL INFORMATION
06.1 Excipients
Each coated tablet contains:
nucleus:
- lactose
- starch
- sodium starch glycolate
- magnesium stearate
coating:
- hypromellose
- titanium dioxide
- macrogol
- talc
06.2 Incompatibility
Not relevant.
06.3 Period of validity
3 years.
06.4 Special precautions for storage
This medicinal product does not require any special storage conditions.
06.5 Nature of the immediate packaging and contents of the package
Cardboard box containing 30 coated tablets packed in PVC and Aluminum blisters.
06.6 Instructions for use and handling
No special instructions.
07.0 MARKETING AUTHORIZATION HOLDER
A. Menarini Industrie Farmaceutiche Riunite s.r.l. - Via Sette Santi 3, 50131 Florence.
08.0 MARKETING AUTHORIZATION NUMBER
A.I.C. n. 023418039
09.0 DATE OF FIRST AUTHORIZATION OR RENEWAL OF THE AUTHORIZATION
Date of first authorization: March 16, 1983
Date of last renewal: May 31, 2005
10.0 DATE OF REVISION OF THE TEXT
AIFA Determination of March 2007