PLEASE NOTE: MEDICINAL PRODUCT NO LONGER AUTHORIZED
What is Trevaclyn?
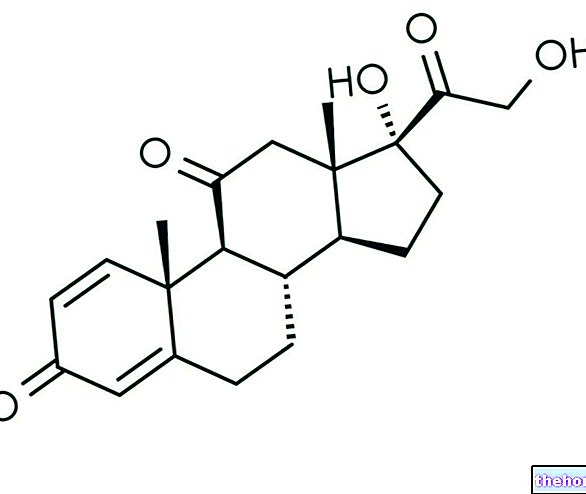
Trevaclyn is a medicine containing two active substances: nicotinic acid (also known as niacin or vitamin B3) and laropiprant. The medicine is available as modified-release tablets. By "modified release" s "we mean that the two active ingredients are released from the tablet at different rates over the course of a few hours.
What is Trevaclyn used for?
Trevaclyn is used as an adjunct to diet and physical activity in patients with dyslipidaemia (exceptionally high levels of fat in the blood), particularly for 'mixed combined dyslipidaemia' and 'primary hypercholesterolaemia'. Patients with mixed combined dyslipidemia have high levels of "bad" cholesterol (LDL) and triglycerides (a type of fat) and low levels of "good" cholesterol (HDL) in their blood. Primary hypercholesterolemia is a condition in which the concentration of cholesterol in the blood is high. By "primary" we mean that hypercholesterolemia has no identifiable cause.
Trevaclyn is normally prescribed together with a statin (the standard medicine used to lower cholesterol) when the efficacy of the statin alone is insufficient. Trevaclyn is used on its own only in patients who cannot take statins.
The medicine can only be obtained with a prescription.
How is Trevaclyn used?
The starting dose of Trevaclyn is one tablet once a day for four weeks; thereafter the dose is increased to two tablets once a day. The medicine is taken orally, with food, in the evening or before going to bed. The tablets must be swallowed whole and must not be divided, broken, crushed or chewed.
Trevaclyn is not recommended for use in children under 18 years of age due to the lack of information on the safety and efficacy profile of the medicine in this group. The medicine should be used with caution in patients with kidney problems and should not be used in patients with liver problems.
How does Trevaclyn work?
The two active substances in Trevaclyn, nicotinic acid and laropiprant, have different mechanisms of action.
Nicotinic acid is a naturally occurring substance that is used in low doses as a vitamin. At higher doses, it reduces the level of fats in the blood through a mechanism that is not yet perfectly
clear. The substance was used for the first time as a medicine capable of modifying the concentration of fats in the blood in the mid-1950s but its use was limited due to side effects, in particular flushing (redness of the skin).
Flushing associated with nicotinic acid is thought to depend on the skin cells releasing a substance called "prostaglandin D2" (PGD2) that dilates (widens) the blood vessels in the skin. Laropiprant blocks the receptors to which the skin normally attaches. PGD2: If the receptors are blocked, PGD2 cannot dilate the vessels in the skin and the frequency and intensity of hot flashes are reduced.
In Trevaclyn tablets, laropiprant is found in one of the layers. The other layer contains nicotinic acid. When the patient takes the tablet, laropiprant is released into the bloodstream first and blocks the PGD2 receptors. Nicotinic acid is released more slowly from the other layer and exerts the action of a drug which modifies the lipid profile.
How has Trevaclyn been studied?
The effects of Trevaclyn were first tested in experimental models before being studied in humans.
Trevaclyn has been studied in four main studies in patients with hypercholesterolaemia or mixed dyslipidaemia.
Two studies looked at Trevaclyn's ability to modify blood fat levels. The first study compared the effectiveness of Trevaclyn with that of nicotinic acid alone or placebo (a dummy treatment) in reducing LDL cholesterol levels in a total of 1,613 patients. The study also looked at the symptoms of hot flashes using a special questionnaire.
The second study compared the combination of Trevaclyn and simvastatin (a statin) with Trevaclyn alone or simvastatin alone in 1,398 patients. The main measure of effectiveness was the change in blood LDL cholesterol levels after 12 weeks.
The third and fourth studies looked at the efficacy of laropiprant in reducing flushing caused by nicotinic acid. They included a total of 2 349 patients taking alternatively Trevaclyn or nicotinic acid. Hot flashes were measured using the Hot Flash Symptoms Questionnaire.
What benefit has Trevaclyn shown during the studies?
Trevaclyn has been shown to be effective in lowering blood LDL cholesterol levels. In the first study, LDL cholesterol levels were reduced by 19% in patients taking Trevaclyn, compared with 1% in those taking placebo. The second study showed that LDL cholesterol levels were further reduced when Trevaclyn was taken together with simvastatin (48% reduction), compared with Trevaclyn alone (17% reduction) or simvastatin alone (37% reduction).
Adding laropiprant to nicotinic acid reduced the symptoms of flushing caused by nicotinic acid. In the first and third studies, fewer patients taking Trevaclyn reported moderate, severe or extreme flushing than patients taking the nicotinic acid only In the fourth study, hot flashes were seen in fewer days in patients taking Trevaclyn than in those taking nicotinic acid alone.
What is the risk associated with Trevaclyn?
The most common side effects with Trevaclyn (seen in more than 1 in 10 patients) are hot flashes. For the full list of side effects reported with Trevaclyn, see the Package Leaflet.
Trevaclyn should not be used in people who may be hypersensitive (allergic) to nicotinic acid, laropiprant or any of the other substances. The medicine should also not be used in patients with liver problems, active stomach ulcer or bleeding. arterial.
Why has Trevaclyn been approved?
The Committee for Medicinal Products for Human Use (CHMP) decided that Trevaclyn's benefits are greater than its risks for the treatment of dyslipidaemia, particularly in patients with mixed combined dyslipidaemia and in patients with primary hypercholesterolaemia. The committee recommended the granting of marketing authorization for Trevaclyn.
Other information about Trevaclyn:
On 3 July 2008, the European Commission granted Merck Sharp & Dohme Ltd. a "Marketing Authorization" for Trevaclyn, valid throughout the European Union.
For the full version of Trevaclyn's EPAR, please click here .
Last update of this summary: 05-2008.
Information about Trevaclyn published on this page may be out of date or incomplete. For a correct use of this information, see the Disclaimer and useful information page.