What is Tafinlar and what is it used for?
Tafinlar is a cancer medicine that contains the active substance dabrafenib. It is used to treat adults with melanoma (a type of skin cancer) that has spread to other parts of the body or is not operable. Tafinlar is only indicated for patients who have been tested for in the genes of melanoma cancer cells of a specific mutation (variation) called “BRAF V600”.
How is Tafinlar used - dabrafenib?
Treatment with Tafinlar should be started and supervised by a doctor experienced in the use of anticancer medicines. The medicine can only be obtained with a prescription. Tafinlar is available as capsules (50 mg and 75 mg). It is given at the recommended dose of 150. mg twice daily, taken at least one hour before a meal or at least two hours after a meal.Treatment should be continued as long as possible, until the disease gets worse or until the side effects become too severe. If the patient experiences certain undesirable effects, it may be necessary to interrupt or discontinue therapy or reduce the dose. For more information, see the Summary of Product Characteristics (also part of the EPAR).
How does Tafinlar work - dabrafenib?
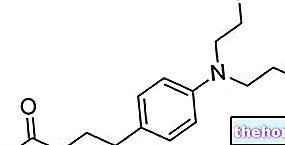
The active substance in Tafinlar, dabrafenib, works by blocking BRAF, a protein that participates in stimulating cell division. In melanomas with the BRAF V600 mutation, there is an abnormal form of BRAF that contributes to tumor development, allowing the uncontrolled division of cancer cells. By blocking the action of the abnormal BRAF protein, Tafinlar helps slow the growth and spread of the tumor. Tafinlar is only given to patients with melanomas caused by the BRAF V600 mutation.
What benefit has Tafinlar - dabrafenib shown during the studies?
Tafinlar has been studied in one main study involving 250 patients with melanoma containing the BRAF V600 mutation that has spread to other parts of the body or is inoperable. Tafinlar was compared with the cancer medicine dacarbazine; the main measure of effectiveness was how long the patients lived until the disease got worse (progression-free survival). In this study, Tafinlar was more effective than dacarbazine in controlling the disease: on average 6 elapsed in patients treated with Tafinlar. , 9 months before the disease got worse compared to 2.7 months in subjects treated with dacarbazine.
What is the risk associated with Tafinlar - dabrafenib?
The most common side effects with Tafinlar (which may affect more than 1 in 10 people) are papilloma (warts), decreased appetite, headache, cough, nausea and vomiting, diarrhea, hyperkeratosis (thickening and hardening of the skin), alopecia (hair loss), rash, hand-foot syndrome (skin reaction and numbness in the palms and soles), arthralgia and myalgia (pain in the joints and muscles), pain in extremities, fever, chills, fatigue and asthenia (a sense of weakness). For the full list of side effects reported with Tafinlar, see the package leaflet
Why has Tafinlar - dabrafenib been approved?
The Agency's Committee for Medicinal Products for Human Use (CHMP) decided that Tafinlar's benefits are greater than its risks and recommended that it be approved for use in the EU. The Committee considered that Tafinlar had convincingly demonstrated to offer clinically relevant benefit to patients with BRAF V600 mutation positive melanoma that has spread to other parts of the body or is inoperable Undesirable effects are considered acceptable and manageable with appropriate measures.
What measures are being taken to ensure the safe and effective use of Tafinlar - dabrafenib?
A risk management plan has been developed to ensure that Tafinlar is used as safely as possible. Based on this plan, safety information has been added to the summary of product characteristics and information leaflet for Tafinlar, including the appropriate precautions to be followed by healthcare professionals and patients.
Other information about Tafinlar - dabrafenib
On 26 August 2013, the European Commission issued a "Marketing Authorization" for Tafinlar, valid throughout the European Union. For more information on Tafinlar therapy, read the package leaflet (included with the EPAR) or consult your doctor. or the pharmacist. Last update of this summary: 08-2013.
The information on Tafinlar - dabrafenib published on this page may be out of date or incomplete. For a correct use of this information, see the Disclaimer and useful information page.