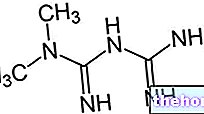
MINIRIN / DDVAP ® is a drug based on Desmopressin
THERAPEUTIC GROUP: Hormones of the posterior lobe of the pituitary

Indications MINIRIN / DDVAP ® - Desmopressin
MINIRIN / DDVAP ® is used in the treatment of diabetes insipidus, primary nocturnal enuresis, post-operative polyuria and nocturia associated with multiple sclerosis.
Desmopressin is also used in the treatment of mild and moderate haemophilia and in some types of Von Willebrand's disease with the aim of increasing concentrations of factor VIII.
Mechanism of action MINIRIN / DDVAP ® - Desmopressin
Desmopressin, active principle of MINIRIN / DDVAP ® is an analogue of vasopressin with which it shares the same biological effects but with a longer duration of action and without vasoconstrictive effects, usually responsible for hypertensive peaks.
From the molecular point of view this molecule is able, in a similar way to vasopressin, to bind specific receptors exposed on the collecting duct, regulating their intracellular activity and facilitating the phosphorylation and consequent translocation on the membrane of a protein known as AQP 2 (aquaporin 2 ), able to facilitate the passage of water from the duct towards the cellular interstitium.
At the same time the aforesaid hormone acts on the cells constituting the ascending tract of the loop of Henle, favoring the reabsorption of sodium and therefore also of water by osmotic gradient.
The biological effects of desmopressin are flanked by particularly advantageous pharmacokinetic properties that allow this molecule to be administered both orally and nasally and parenterally injected.
Studies carried out and clinical efficacy
1. THE EFFECTIVENESS OF DESMOPRESSIN IN THE TREATMENT OF NICTURIA IN ELDERLY PATIENTS
J Res Med Sci. 2011 Apr; 16: 516-23.
Efficacy of desmopressin in treatment of nocturia in elderly men.
Rezakhaniha B, Arianpour N, Siroosbakhat S.
Double-blind clinical trial demonstrating how oral administration of Desmopressin, before going to bed, can be effective in reducing nocturia in elderly male patients.
2. DESMOPRESSIN AND BEHAVIORAL ASPECTS IN THE MANAGEMENT OF NIGHT ENURESIS.
Urol Nurs. 2011 Sep-Oct; 31: 286-9.
Desmopressin versus behavioral modifications as initial treatment of primary nocturnal enuresis.
Fera P, Lelis MA, Glashan Rde Q, Pereira SG, Bruschini H.
Work that reaffirms the importance of behavioral aspects in the initial treatment of primary nocturnal enuresis. The results obtained, if started promptly, seem to approach those achieved with drug therapy based on desmopressin.
3. DESMOPRESSIN IN KIDNEY TRANSPLANT
Transplantation. 2011 Dec 15; 92: 1252-8.
Donor desmopressin is associated with superior graft survival after kidney transplantation.
Benck U, Gottmann U, Hoeger S, Lammert A, Rose D, Boesebeck D, Lauchart W, Birck R, Weiss C, Krämer BK, Yard BA, Schnuelle P.
Important study that demonstrates how pretreatment of brain death with dopamine and desmopressin can limit kidney injury during transplantation, improving survival and functionality of the transplanted organ.
Method of use and dosage
MINIRIN / DDVAP ®
Tablets of 0.1 - 0.2 mg of Desmopressin acetate;
Sublingual tablets of 60 - 120 mcg of Desmopressin acetate;
Nasal spray of 0.125 mg of Desmopressin acetate;
Injectable preparation of 4 mcg of Desmopressin cetate;
Desmopressin Acetate 250mcg Oral Drops:
the dosage and the dosage schedule vary considerably from patient to patient according to the therapeutic needs, the physio-pathological conditions and the pharmaceutical format used.
The doses used could undergo even significant variations based on clinical efficacy or on the possible appearance of side effects.
Warnings MINIRIN / DDVAP ® - Desmopressin
The use of MINIRIN / DDVAP ® must be preceded by a careful medical examination useful for assessing the patient's state of health and prescribing appropriateness.
It is useful for the doctor to remind the patient to significantly reduce the quantity of liquids to be ingested during therapy in order to avoid potential water intoxication, responsible for even serious side effects.
At the same time, the doctor must monitor the patient's state of health, the correct functioning of the urinary tract and the blood sodium concentrations throughout the therapeutic process, especially in elderly patients, in order to reduce the risk of hyponatremia.
MINIRIN / DDVAP ® contains lactose therefore it is not suitable for subjects suffering from glucose / galactose malabsorption syndrome, lactase enzyme deficiency or galactosemia.
PREGNANCY AND BREASTFEEDING
Although the studies present at the moment do not seem to report side effects on the fetus related to the intake of desmopressin in the pregnant woman, the absence of statistically significant data does not allow the free use of these drugs during pregnancy.
Interactions
The particular biological activity of desmopressin could be responsible for clinically relevant drug interactions.
More precisely, the contextual intake of loperamide and analgesics could cause an increase in the blood concentrations of desmopressin when taken orally, with a consequent accentuation of the biological effects of the drug.
The concomitant intake of tricyclic antidepressants, serotonin re-uptake inhibitors, carbamazepine and chlorpromazine could increase the risk of water retention and hyponatremia.
Contraindications MINIRIN / DDVAP ® - Desmopressin
The use of MINIRIN / DDVAP ® is contraindicated in patients suffering from polyuria associated with habitual or psychogenic polydipsia, established heart failure, moderate and severe renal insufficiency, hyponatremia, syndrome of inappropriate ADH secretion and in case of hypersensitivity to the active substance or to one of its excipients.
Undesirable Effects - Side Effects
Different clinical trials and a careful post-marketing monitoring have shown how the assumption of MINIRIN / DDVAP ® according to the indications of the specialist doctor can be devoid of significant side effects.
The most frequently observed adverse reactions were headache, abdominal pain and nausea.
However, it is useful to remember how it becomes necessary to reduce the ingestion of liquids during treatment with MINIRIN / DDVAP ® in order to avoid water retention and hyponatremia often accompanied by headaches, nausea, weight gain and, in severe cases, convulsions.
Note
MINIRIN / DDVAP ® can only be sold under medical prescription
The information on MINIRIN / DDVAP ® - Desmopressin published on this page may be out of date or incomplete. For a correct use of this information, see the Disclaimer and useful information page.


















.jpg)