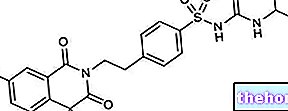
AVANDIA ® a drug based on Rosiglitazone.
THERAPEUTIC GROUP: Oral hypoglycemic agents - Thiazolidinediones

Indications AVANDIA ® - Rosiglitazone
AVANDIA ® is a hypoglycemic drug used in the treatment of type II diabetes, not adequately compensated by non-pharmacological therapeutic measures such as diet and physical activity.
This medicine can also be used successfully in combination therapy with metformin and sulfonylureas, when the latter are ineffective at the maximum tolerated dose.
Mechanism of action AVANDIA ® - Rosiglitazone
AVANDIA ® is a rosiglitazone-based drug, an active ingredient belonging to the thiazolidinediones family, capable of acting at the level of insulin-dependent peripheral tissues, improving their sensitivity to insulin, and supporting the important hypoglycemic effect.
The therapeutic effect of rosiglitazone is essentially associated with the ability to act as a selective agonist of PPAR gamma receptors, fundamental from a biological point of view, as they are involved in the modulation of the expression of genes active on glucose, lipid and inflammatory metabolism, and expressed precisely at the level of the main insulin-sensitive peripheral tissues.
From a pharmacokinetic point of view, on the other hand, oral intake of rosiglitazone precedes intestinal absorption with a bioavailability of 99%, hepatic biotransformation and subsequent renal elimination of inactive metabolites.
Clinical studies show how the hypoglycemic effect can take place both in basal and post-prandial conditions, guaranteeing in a few weeks of treatment, a significant reduction in the concentrations of glycosylated hemoglobin, an important marker of glycemic control.
Studies carried out and clinical efficacy
1. ROSIGLITAZONE, WITHDRAWAL FROM THE MARKET
Rev Med Liege. 2010 Oct; 65: 574-9.
Suspension of the commercialization of sibutramine and rosiglitazone in Europe.
Scheen AJ.
In 2010, the EMEA suspended the marketing of rosiglitazone throughout the European territory, given the significant increase in cardiovascular risk, especially myocardial ischemia, in patients treated with this active ingredient.
2. ROSIGLITAZONE AND CARDIOVASCULAR RISK
Curr Atheroscler Rep. 2008 Oct; 10: 398-404.
Rosiglitazone and cardiovascular risk.
Kaul S, Diamond GA.
The re-reading of 42 different clinical trials that evaluated the efficacy and side effects of rosiglitazone, showed that therapy with this active ingredient was associated with a 43% increase in the risk of myocardial infarction and 64% of deaths due to of cardiovascular pathologies.
3. ROSIGLITAZONE, OPEN DEBATE
Diabet Med. 2010 Dec; 27: 1392-400.
The effects of rosiglitazone on atherosclerotic progression in patients with Type 2 diabetes at high cardiovascular risk.
Yee MS, Pavitt DV, Dhanjil S, Godsland IF, Richmond W, Johnston DG.
Although the EMEA has come to the conclusion to suspend the marketing of rosiglitazone-based products, the debate on potential side effects and the real risk remains open. A significant example comes from this recent study, which demonstrates how second type diabetic patients, treated with rosiglitazone, show a decrease in the thickness of the carotid atheromatous plaque, with a significant improvement in glycemic control, insulin sensitivity and fibrinolytic action
Method of use and dosage
AVANDIA ® 4 mg rosiglitazone tablets:
the most used dosage was that of 4 mg per day, possibly to be increased to 8 mg / day, in case of therapeutic failure, to be taken with a glass of water regardless of meals.
It is important to consider that in the case of combined therapy, it is of fundamental importance to start treatment with the lowest effective doses.
Warnings AVANDIA ® - Rosiglitazone
AVANDIA ® was withdrawn from the market in 2010, given the high risks that do not justify the potential benefits derived from the use of this medicine.
In fact, the important alterations of the hydro-electrolytic balance induced by rosiglitazone significantly accentuate the appearance of signs, symptoms or episodes triggering important cardiac pathologies such as myocardial insufficiency or ischemia.
Furthermore, the risk of bone pathologies, of significant alterations in liver function, and the risks of hypoglycemia, required continuous monitoring of the haematochemical, functional and metabolic picture, in order to periodically check the patient's state of health.
PREGNANCY AND BREASTFEEDING
The ability of rosiglitazone to cross the placental filter and to find itself in breast milk, altering the normal evolutionary and metabolic balances of the fetus and infant, push the experts in the field to strongly contraindicate the use of this active ingredient in the treatment of gestational diabetes.
Interactions
The pharmacokinetic properties of rosiglitazone can be significantly modified by the concomitant intake of gemfibrozil, rifamipicin and other modulators of the hepatic enzyme responsible for its CYP2C8 metabolism, such as phenytoin, carbamazepine, phenobarbital, Hypericum Perforatum.
It should be remembered that dual or triple therapy, with other hypoglycemic drugs, could significantly accentuate the hypoglycemic effects of rosiglitazone.
Contraindications AVANDIA ® - Rosiglitazone
AVANDIA ® contraindicated in case of known hypersensitivity to the active substance or to one of its excipients, heart failure and associated episodes, acute coronary syndrome, impaired liver and kidney function, diabetic keto acidosis and diabetic pre-coma.
The use of AVANDIA ® is strongly discouraged even during the period of pregnancy and breastfeeding.
Undesirable Effects - Side Effects
Rosiglitazone treatment has been subjected to dozens of clinical studies given the numerous potential side effects, such as to make its use particularly dangerous.
In fact, in addition to the common side effects, also described for other hypoglycemic drugs, such as the risk of hypoglycemia, metabolic and nutritional alterations, alterations in the haematological picture, gastro-intestinal disorders and alterations in the hydro-electrolyte balance, treatment with AVANDIA ® exposes the patient at a high risk of heart disease such as heart failure and ischaemia.
In fact, this last condition seems to be responsible for the withdrawal from the market of all rosiglitazone-based drugs, given the low BENEFITS / RISKS ratio.
Note
THE AVANDIA DRUG HAS BEEN WITHDRAWN FROM THE EUROPEAN MARKET.
The information on AVANDIA ® - Rosiglitazone published on this page may be out of date or incomplete. For a correct use of this information, see the Disclaimer and useful information page.