Active ingredients: Deflazacort
FLANTADIN 6 mg tablets
FLANTADIN 30 mg tablets
FLANTADIN 22.75 mg / ml oral drops, suspension
Indications Why is Flantadin used? What is it for?
PHARMACOTHERAPEUTIC CATEGORY
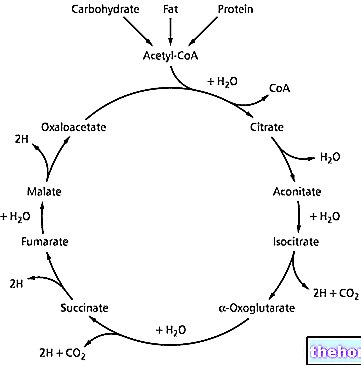
Deflazacort is a synthetic glucocorticoid with anti-inflammatory and immunosuppressive activity.
THERAPEUTIC INDICATIONS
Insufficient primary and secondary activity of the adrenal glands (alone or in association with mineralocorticoids). Rheumatic diseases: psoriatic arthropathy, rheumatoid arthritis, ankylosing spondylitis, acute gouty arthropathy, post-traumatic osteoarthritis, acute and subacute bursitis, acute nonspecific tenosynovitis, epicondylitis. Collagen diseases: systemic lupus erythematosus (SLE), acute rheumatic carditis (cardiac rheumatism), systemic dermatomyositis (polymyositis). Dermatological diseases: pemphigus, bullous herpetiform dermatitis, severe polymorphic erythema (Stevens-Johnson syndrome), exfoliative dermatitis, mycosis fungoides (cutaneous lymphoma), severe psoriasis, severe seborrheic dermatitis. Allergic states: seasonal or permanent allergic rhinitis, bronchial asthma, contact dermatitis, atopic dermatitis, serum sickness, drug hyperreactivity. Respiratory diseases: symptomatic sarcoidosis, berylliosis, fulminant or disseminated pulmonary tuberculosis (in association with appropriate chemotherapy), aspiration pneumonia. Ocular diseases (severe, acute and chronic inflammatory and allergic processes): allergic marginal corneal ulcers, ocular herpes zoster, inflammation of the anterior segment of the globe, choroiditis and diffuse posterior uveitis, sympathetic ophthalmitis, allergic conjunctivitis, keratitis, chorioretinitis, neuritis of the " optic, iritis, and iridocyclitis Haematological disorders and haematological diseases with malignant evolution: secondary thrombocytopenia of the adult, autoimmune haemolytic anemia, erythroblastopenia, congenital hypoplastic anemia; Hodgkin's disease, non-Hodgkin's lymphomas, chronic lymphatic leukemia, acute childhood leukemia, etc. Edematous states: idiopathic nephrotic syndrome or secondary to SLE Gastrointestinal diseases: ulcerative colitis, regional enteritis.
Contraindications When Flantadin should not be used
Active tuberculosis, peptic ulcer, ocular herpes simplex, systemic fungal infections, psychosis. Generally contraindicated in pregnancy, lactation and very early childhood (see also "Special Warnings"). Hypersensitivity to the active substance or to any of the excipients.
Precautions for use What you need to know before taking Flantadin
In patients on corticosteroid therapy, subjected to particular stress, it is essential to adjust the dose in relation to the entity of the stressful condition.
During prolonged therapy and with high doses, should an alteration of the electrolyte balance occur, it is advisable to adjust the sodium and potassium intake. Corticosteroids increase the excretion of calcium.
A state of secondary adrenal insufficiency, induced by cortisone, can be contained with a gradual reduction of doses. This type of relative insufficiency can persist for months after discontinuation of therapy. Therefore in any stressful situation, which occurs during this period, adequate hormone therapy should be instituted. In this situation the mineralocorticoid secretion can be compromised and therefore it would be advisable to administer, concomitantly, salt and / or mineralocorticoids.
In patients with hypothyroidism or with cirrhosis of the liver, the response to corticosteroids may be increased.
The use of FLANTADIN in active tuberculosis should be limited to cases of fulminant or disseminated disease, in which the corticosteroid must be used with appropriate antituberculous therapy. If corticosteroids are administered to patients with latent tuberculosis or with a positive response to tuberculin, close surveillance as disease activation may occur. In prolonged corticotherapy these patients should receive chemoprophylaxis.
Corticosteroids should be administered with caution in the following cases: non-specific ulcerative colitis with danger of perforation, abscesses and pyogenic infections in general, diverticulitis, recent intestinal anastomosis, renal failure, hypertension, diabetes, osteoporosis, myasthenia gravis.
Children undergoing prolonged corticotherapy must be closely monitored from the point of view of growth and development.
Interactions Which drugs or foods can modify the effect of Flantadin
Although there are no known interactions with other medicinal products and incompatibility with FLANTADIN, during concomitant treatment with anticonvulsants (phenobarbital, diphenylhydantoin), some antibiotics (rifampicin), anticoagulants (warfarin) or bronchodilators (ephedrine), it is suggested to increase the maintenance dose. of the glucocorticoid. During concomitant treatment with other antibiotics (erythromycin, troleandomycin), estrogen or preparations containing estrogen, it is recommended to reduce the glucocorticoid dose.
In patients with hypoprothrombinemia, caution is advised in combining acetylsalicylic acid with corticosteroids.
Antacids administered simultaneously to decrease the dyspepsia they induce reduce intestinal absorption of glucocorticoids, worsening the control of disease symptoms.
Tell your doctor or pharmacist if you have recently taken any other medicines, even those without a prescription.
Warnings It is important to know that:
The tablets contain lactose and the 6 mg tablets also contain sucrose, while the oral suspension contains sorbitol, so in case of ascertained intolerance to sugars contact your doctor before taking the medicine. Corticosteroids can mask some signs of infection, and intercurrent infections can occur during their use. In these cases, the opportunity to institute adequate antibiotic therapy must always be evaluated.
Patients on corticosteroid therapy should not be vaccinated.
Psychic alterations of various kinds can occur during corticotherapy: euphoria, insomnia, changes in mood or personality, severe depression or symptoms of real psychosis. A pre-existing emotional instability or psychotic tendencies can be aggravated by corticosteroids.
Pregnancy and breastfeeding
In pregnant women, during lactation and in very early childhood, the product should be administered in cases of real need, under the direct supervision of the doctor.
Effects on ability to drive and use machines
The effects of glucocorticoids in this regard are not known.
For those who carry out sporting activities: the use of the drug without therapeutic necessity constitutes doping and can in any case determine positive anti-doping tests.
Dose, Method and Time of Administration How to use Flantadin: Posology
FLANTADIN is a medicine that is to be administered orally. The initial daily dosage in adults can vary from 6 to 90 mg (one or more tablets or more drops per day), in consideration of the severity and evolution of the specific disease to be treated.
The starting dosage should be maintained or modified until a satisfactory clinical response is achieved. It is important to underline that the corticosteroid requirement is variable and therefore the posology must be individualized taking into account the disease and the patient's therapeutic response.
The maintenance dosage must always be the minimum capable of controlling the symptoms: the dosage reduction must always be done gradually.
With regard to the presentation in drops, it should be noted that the dropper of the suspension delivers on average 1 mg of deflazacort per drop.
IT IS SUGGESTED TO SHAKE THE BOTTLE BEFORE USE AND TO DILUTE THE SUSPENSION, IMMEDIATELY BEFORE ADMINISTRATION, IN SUGAR WATER OR IN DRINKS NOT ADDED WITH CARBON DIOXIDE.
It is advisable to take the daily dose of FLANTADIN in a single administration, in the morning, together with small quantities of food.
Overdose What to do if you have taken too much Flantadin
In case of overdose it is recommended to carry out, in concomitance with the usual measures for the elimination of the unabsorbed drug (gastric lavage, charcoal, etc.), a clinical check of the patient's vital functions.
In case of accidental intake of an excessive dose of the medicine, notify your doctor immediately or go to the nearest hospital.
If you have any further questions on the use of FLANTADIN, ask your doctor or pharmacist.
Side Effects What are the side effects of Flantadin
It should be borne in mind that during corticosteroid therapy, especially for intense and prolonged treatments, some of the following effects may occur:
- alterations in the hydroelectrolytic balance which, rarely and in particularly predisposed patients, can lead to hypertension and congestive heart failure;
- musculoskeletal changes, such as osteoporosis, bone fragility, myopathies;
- complications affecting the gastrointestinal system, which can lead to the appearance or activation of a peptic ulcer;
- skin alterations, such as delays in the healing process, thinning and fragility of the skin;
- neurological changes such as dizziness, headache and increased intracranial pressure;
- interference with the functionality of the pituitary-adrenal axis, particularly in times of stress; alteration of endocrine function, such as menstrual irregularities, changes in physiognomy ("moon face"), growth disturbances in children, decreased sugar tolerance with the possible occurrence of latent diabetes mellitus as well as an increased need for antidiabetic drugs, to be determined in the opinion of the doctor;
- complications of an ophthalmological nature such as posterior subcapsular cataract and increased intraocular pressure;
- negativization of the nitrogen balance.
Compliance with the instructions contained in the package leaflet reduces the risk of undesirable effects. Like all medicines, FLANTADIN can cause side effects, although not everybody gets them.
If any of the side effects gets serious or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
Expiry and Retention
Expiry: see the expiry date indicated on the package.
WARNING: do not use the medicine after the expiry date indicated on the package. The expiry date indicated refers to the product in intact packaging, correctly stored.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. This will help protect the environment.
KEEP THE MEDICINAL PRODUCT OUT OF THE REACH AND SIGHT OF CHILDREN
COMPOSITION
Flantadin 6 mg tablets
One tablet contains:
Active ingredient: Deflazacort 6 mg. Excipients: Lactose; Magnesium stearate; Cornstarch; Microcrystalline cellulose; Sucrose.
Flantadin 30 mg tablets
One tablet contains:
Active ingredient: Deflazacort 30 mg. Excipients: Lactose; Magnesium stearate; Cornstarch; Microcrystalline cellulose
Flantadin 22.75 mg / ml oral drops, suspension 1 ml suspension contains:
Active principle:
Deflazacort 22.75 mg. Excipients: Aluminum and magnesium silicate; Sodium carboxymethylcellulose; Benzyl alcohol; Sorbitol; Polysorbate 80; Acetic acid; Purified water.
PHARMACEUTICAL FORMS AND CONTENT
Box of 10 tablets of 6 mg in blister.
Box of 10 tablets of 30 mg in blister.
Oral suspension: bottle of 13 ml of suspension, with dropper.
Source Package Leaflet: AIFA (Italian Medicines Agency). Content published in January 2016. The information present may not be up-to-date.
To have access to the most up-to-date version, it is advisable to access the AIFA (Italian Medicines Agency) website. Disclaimer and useful information.
01.0 NAME OF THE MEDICINAL PRODUCT
FLANTADIN
02.0 QUALITATIVE AND QUANTITATIVE COMPOSITION
• Flantadin 6 mg tablets
One tablet contains:
Active ingredient: Deflazacort 6 mg.
• Flantadin 30 mg tablets
One tablet contains:
Active ingredient: Deflazacort 30 mg.
• Flantadin 22.75 mg / ml oral drops, suspension
1 ml of suspension contains:
Active ingredient: Deflazacort 22.75 mg.
For the full list of excipients, see 6.1.
03.0 PHARMACEUTICAL FORM
Tablets - oral drops, suspension.
04.0 CLINICAL INFORMATION
04.1 Therapeutic indications
Primary and secondary adrenocortical insufficiency (alone or in association with mineralocorticoids). Rheumatic diseases: psoriatic arthropathy, rheumatoid arthritis, ankylosing spondylitis, acute gouty arthropathy, post-traumatic osteoarthritis, acute and subacute bursitis, acute nonspecific tenosynovitis, epicondylitis. Collagen diseases: systemic lupus erythematosus (SLE), acute rheumatic carditis (cardiac rheumatism), systemic dermatomyositis (polymyositis). Dermatological diseases: pemphigus, bullous herpetiform dermatitis, severe polymorphic erythema (Stevens-Johnson syndrome), exfoliative dermatitis, mycosis fungoides (cutaneous lymphoma), severe psoriasis, severe seborrheic dermatitis. Allergic states: seasonal or permanent allergic rhinitis, bronchial asthma, contact dermatitis, atopic dermatitis, serum sickness, drug hyperreactivity. Respiratory diseases: symptomatic sarcoidosis, berylliosis, fulminant or disseminated pulmonary tuberculosis (in association with appropriate chemotherapy), aspiration pneumonia. Ocular diseases (severe, acute and chronic inflammatory and allergic processes): allergic marginal corneal ulcers, ocular herpes zooster, inflammation of the anterior segment of the eyeball, choroiditis and posterior diffuse uveitis, sympathetic ophthalmitis, allergic conjunctivitis, keratitis, chorioretinitis, neuritis of the " optic, iritis, and iridocyclitis.
Haematological disorders and haematological diseases with malignant evolution: secondary thrombocytopenia in adults, autoimmune hemolytic anemia, erythroblastopenia, congenital hypoplastic anemia; Hodgkin's disease, non-Hodgkin's lymphomas, chronic lymphatic leukemia, acute childhood leukemia, etc. Edematous states: idiopathic nephrotic syndrome or secondary to SLE. Gastrointestinal diseases: ulcerative colitis, regional enteritis.
04.2 Posology and method of administration
The initial daily dosage in adults can vary from 6 to 90 mg, in consideration of the severity and evolution of the specific disease to be treated.
The starting dosage should be maintained or modified until a satisfactory clinical response is achieved. It is important to underline that the corticosteroid requirement is variable and therefore the posology must be individualized taking into account the disease and the patient's therapeutic response.
The maintenance dosage must always be the minimum capable of controlling the symptoms, minimizing the risk of secondary effects: the dosage reduction must always be implemented gradually.
Regarding the presentation in drops, it should be noted that the dropper of the suspension delivers on average 1 mg of Deflazacort per drop.
IT IS SUGGESTED TO SHAKE THE BOTTLE BEFORE USE AND TO DILUTE THE SUSPENSION, IMMEDIATELY BEFORE ADMINISTRATION, IN SUGAR WATER OR IN DRINKS NOT ADDED WITH CARBON DIOXIDE.
It is advisable to take the daily dose of FLANTADIN in a single administration, in the morning, together with small quantities of food.
04.3 Contraindications
Active tuberculosis, peptic ulcer, ocular herpes simplex, systemic fungal infections, psychosis. Generally contraindicated in pregnancy, lactation and very early childhood (see section 4.6).
Hypersensitivity to the active substance or to any of the excipients.
04.4 Special warnings and appropriate precautions for use
In patients on corticosteroid therapy, subjected to particular stress, it is essential to adapt the dose of the glucocorticoid in relation to the extent of the stressful condition. Corticosteroids can mask some signs of infection and during their use intercurrent infections may occur (opportunity to establish antibiotic therapy). A state of secondary adrenal insufficiency, induced by cortisone, can be contained with a gradual reduction of doses. This type of relative insufficiency can persist for months after the discontinuation of therapy. Therefore in any stressful situation, which is manifested in this period, an adequate hormone replacement therapy should be instituted.In this situation the mineralocorticoid secretion may be compromised and therefore it would be advisable to administer, concomitantly, salt and / or mineralocorticoids.In patients with hypothyroidism or with cirrhosis of the liver, the response to corticosteroids may be increased.
Psychic alterations of various kinds can occur during corticotherapy: euphoria, insomnia, changes in mood or personality, severe depression or symptoms of real psychosis. A pre-existing emotional instability or psychotic tendencies can be aggravated by corticosteroids.
Patients on corticosteroid therapy should not be vaccinated against smallpox. Other immunizing procedures should not be undertaken in patients receiving corticosteroids especially at high doses, due to increased risk of neurological complications and decreased antibody response.
The use of Flantadin in active tuberculosis should be limited to cases of fulminant or disseminated disease, in which the corticosteroid is used with appropriate antituberculous therapy. If corticosteroids are administered to patients with latent tuberculosis or with a positive response to tuberculin, close surveillance as disease activation may occur. In prolonged corticotherapy these patients should receive chemoprophylaxis.
Corticosteroids should be administered with caution in the following cases: non-specific ulcerative colitis with danger of perforation, abscesses and pyogenic infections in general, diverticulitis, recent intestinal anastomosis, renal failure, hypertension, diabetes, osteoporosis, myasthenia gravis. Children undergoing prolonged corticotherapy must be closely monitored from the point of view of growth and development.
In case of concomitant treatment with diuretics (thiazides, furosemide, etc.) and beta 2 agonists (reproterol, etc.) which induce potassium loss, check potassium and blood pH.
The tablets contain lactose and the 6 mg tablets also contain sucrose, so patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency, or glucose-galactose malabsorption should not take this medicine.
The oral suspension contains sorbitol therefore patients with rare hereditary problems of fructose intolerance should not take this medicine.
04.5 Interactions with other medicinal products and other forms of interaction
Although there are no known interactions with other medicinal products and incompatibility with Flantadin, however during simultaneous treatment with: anticonvulsants (phenobarbital, diphenylhydantoin), some antibiotics (rifampicin), anticoagulants (warfarin), bronchodilators (ephedrine) it is suggested to increase the dose of maintenance of glucocorticoid. During concomitant treatment with other antibiotics (erythromycin, troleandomycin), estrogen or preparations containing estrogen, it is recommended to reduce the glucocorticoid dose.
In patients with hypoprothrombinemia, caution is advised in combining acetylsalicylic acid with corticosteroids.
Antacids administered simultaneously to decrease the dyspepsia they induce reduce intestinal absorption of glucocorticoids, worsening the control of disease symptoms.
04.6 Pregnancy and lactation
In pregnant, lactating and early childhood women, the product should be administered in cases of real need, under the direct supervision of the doctor.
04.7 Effects on ability to drive and use machines
The effects of glucocorticoids in this regard are not known.
04.8 Undesirable effects
It should be borne in mind that during corticosteroid therapy, especially for intense and prolonged treatments, some of the following effects may occur:
- alterations in the hydroelectrolytic balance which, rarely and in particularly predisposed patients, can lead to hypertension and congestive heart failure;
- musculoskeletal alterations, such as osteoporosis, bone fragility, myopathies;
- complications affecting the gastrointestinal system, which can lead to the appearance or activation of a peptic ulcer;
- skin alterations, such as delays in the healing process, thinning and fragility of the skin;
- neurological alterations such as dizziness, headache and increased intracranial pressure;
- interference with the functionality of the pituitary-adrenal axis, particularly in times of stress; dysendocrinies, such as menstrual irregularities, shushingoid-like appearance, growth disturbances in children, decreased tolerance to carbohydrates with possible manifestation of latent diabetes mellitus as well as an increased need for hypoglycemic drugs in diabetics, to be determined in the opinion of the doctor;
- ophthalmic complications such as posterior subcapsular cataract and increased intraocular pressure;
- negativization of the nitrogen balance.
04.9 Overdose
During prolonged therapy and with high doses, should an alteration of the electrolyte balance occur, it is advisable to adjust the sodium and potassium intake. Corticosteroids increase urinary excretion of calcium.
In case of overdose it is recommended to carry out, in concomitance with the usual measures for the elimination of the unabsorbed drug (gastric lavage, charcoal, etc.), clinical monitoring of the patient's vital functions.
05.0 PHARMACOLOGICAL PROPERTIES
05.1 Pharmacodynamic properties
Pharmacotherapeutic group: Systemic corticosteroids, unassociated: glucocorticoids ATC code: H02AB13
The study of different experimental models indicates that Flantadin is an effective inhibitor of the early exudative phase of inflammation (edema induced by carrageenan and nystatin), as well as the formation of slowly evolving granulomatous inflammatory tissue (granuloma from cotton pellets). also shown to inhibit experimentally induced chronic (joint) inflammatory manifestations (adjuvant arthritis). The study of the ability of glucocorticoids to induce glycogen storage in the liver of adrenalectomized rats, showed that Flantadin is able to produce an increase in glyconeogenesis and hepatic glycogenosynthesis about 10 times that of prednisolone administered in equally active doses. The anti-inflammatory potency of Flantadin, estimated on the basis of these well-established experimental models, is about 10-20 times that of prednisolone or 40 times that of cortisol (hydrocortisone), while its duration of anti-inflammatory effects is greater hours of that of other glucocorticoids administered in equivocal doses (prednisolone, triamcinolone, etc.).
The study of the ability of glucocorticoids to induce a reduction in renal excretion of Na + in adrenalectomized animals (mineralocorticoid effect), showed that Flantadin did not induce, unlike a typical mineralocorticoid hormone such as DOCA, tissue retention of Na +, while causing like prednisolone an "increased renal excretion of fluids and K +.
The study of the ability of glucocorticoids to induce fasting and post-glucose hyperglycemia showed that Flantadin administered orally in rats induced fasting and post-glucose hyperglycemic levels comparable to those produced by prednisolone, while intraperitoneally produced a reduction in glucose tolerance after loading, significantly lower than that induced by prednisolone in equactive doses. The study of the secondary effects of Flantadin at the level of other systems and apparatuses, has shown that it interferes in an irrelevant way on the central nervous and cardiovascular systems during repeated administration (a few days) in the rat.
05.2 "Pharmacokinetic properties
The study of the pharmacokinetics, tissue distribution and metabolism of Flantadin was carried out in rats, guinea pigs, monkeys and humans, using methods of analytical determination on the compound as such and on the labeled one (C14). After being rapidly absorbed from the intestine (plasma peak between 1 and 2 hours), Deflazacort is immediately hydrolyzed into its metabolites, 21-desacetyl Deflazacort (the main or active metabolite II) and 6-beta hydroxy derivative (or metabolite III inactive), as there are no more traces of the compound as such in the circulation (pro-drug). The active metabolites of Flantadin then follow the same metabolic fate as prednisolone and other synthetic glucocorticoids. The plasma half-life of metabolite II ranges from 2 hours in humans to 4 hours in monkeys. The study of the tissue distribution of the labeled drug in the rat, having revealed that its preferential "target organs" are the kidney and blood cells, suggests that the longer persistence of the drug in these compartments is responsible for its longer duration of effects. The elimination of metabolites occurs in humans within 24 hours, mainly through the urine.
05.3 Preclinical safety data
The study of the effects deriving from the administration of single doses indicates that the LD50 are: after oral administration 5200 mg / kg in the mouse and> 4000 mg / kg in the dog; after SC administration 1610 mg / kg in the mouse, 109 mg / kg in the rat and 50 mg / kg in the dog.
The study of the effects resulting from repeated oral administration in the rat (1.75 - 7.0 mg / kg / day), dog (0.1 - 1 mg / kg / day) and monkey (0.5 - 1, 5 mg / kg / day), lasting 6-12 months, showed that Flantadin is satisfactorily tolerated, with secondary effects affecting organs, comparable to those usually detected with other glucocorticoids, under the same experimental conditions.
The study of the effects on reproduction (fertility, embryotoxicity and peripostnatal) showed that Flantadin induced secondary alterations comparable to those usually observed with other glucocorticoids, under the same experimental conditions. Flantadin has never produced mutagenic effects.
Carcinogenicity studies conducted in rodents have shown no tumorigenic effect in mice, while in rats some neoplastic effects similar to those already known for other corticosteroids have been observed, without any confirmation with the use of these compounds in humans.
06.0 PHARMACEUTICAL INFORMATION
06.1 Excipients
• Flantadin - 6 mg tablets
Lactose, Magnesium stearate, Corn starch, Microcrystalline cellulose, Sucrose.
• Flantadin - 30 mg tablets
Lactose, Magnesium stearate, Corn starch, Microcrystalline cellulose.
• Flantadin - 22.75 mg / ml oral drops, suspension
Aluminum and magnesium silicate, Sodium carboxymethylcellulose, Benzyl alcohol, Sorbitol 70% solution, Polysorbate 80, Acetic acid to taste at pH 4, purified water.
06.2 Incompatibility
Patients on corticosteroid therapy should not be vaccinated against smallpox. Other immunizing procedures should not be undertaken in patients receiving corticosteroids especially at high doses, due to increased risk of neurological complications and decreased antibody response.
Antacids administered simultaneously to decrease the dyspepsia they induce reduce intestinal absorption of glucocorticoids, worsening the control of disease symptoms.
06.3 Period of validity
Tablets: 5 years
Drops: 3 years
06.4 Special precautions for storage
This medicine does not require any special storage conditions.
06.5 Nature of the immediate packaging and contents of the package
- Box of 10 tablets of 6 mg in blister
- Box of 10 tablets of 30 mg in blister
- Oral suspension: bottle of 13 ml of suspension, with dropper
06.6 Instructions for use and handling
It is recommended to take it in the morning on a full stomach. It is important to remember to shake the bottle of the oral suspension well before use.
07.0 MARKETING AUTHORIZATION HOLDER
TEOFARMA S.r.l.
Via F.lli Cervi, 8
27010 Salimbene Valley (PV)
08.0 MARKETING AUTHORIZATION NUMBER
• Flantadin 6 mg tablets: 10 tablets - A.I.C. n. 025464037
• Flantadin 30 mg tablets: 10 tablets - A.I.C. n. 025464049
• Flantadin 22.75 mg / ml oral drops, suspension - 13 ml bottle - A.I.C. n. 025464052
09.0 DATE OF FIRST AUTHORIZATION OR RENEWAL OF THE AUTHORIZATION
• Flantadin 6 mg tablets: 10 tablets - 6.4.1985 / 31.5.2010
• Flantadin 30 mg tablets: 10 tablets - 6.4.1985 / 31.5.2010
• Flantadin 22.75 mg / ml oral drops, suspension - 13 ml bottle - 5.9.1994 / 31.5.2010
10.0 DATE OF REVISION OF THE TEXT
June 2010