Fats or Lipids
Lipids are ternary organic substances insoluble in water and soluble in non-polar solvents such as ether and benzene.
From a nutritional point of view they are divided into:
- DEPOSIT LIPIDS (98%), with energy function (triglycerides);
- CELLULAR LIPIDS (2%), with structural function (phospholipids, glycolipids, cholesterol).
From a chemical point of view they are divided into:
- SAPONIFIABLE OR COMPLEX: they can be broken down, by hydrolysis, into fatty acids and molecules carrying one or more alcohol groups (glycerides, phospholipids, glycolipids, waxes, sterides);
- NOT SAPONIFIABLE OR SIMPLE: they do not contain fatty acids in their structure (terpenes, steroids, prostaglandins).
In the human organism and in the foods that nourish it, the most abundant lipids are triglycerides (or triacylglycerols). They are formed by the union of three fatty acids with a molecule of glycerol.

LEGEND:

The functional group of an organic molecule consisting of an oxygen atom bonded with a double bond to a carbon atom that is also bonded to a hydroxyl group (-OH) is called a carboxylic group.
Fatty acids
Fatty acids, fundamental components of lipids, are molecules made up of a chain of carbon atoms, called aliphatic chain, with a single carboxylic group (-COOH) at one end. The aliphatic chain that constitutes them tends to be linear and only in rare cases occurs in a branched or cyclic form. The length of this chain is extremely important, as it influences the physico-chemical characteristics of the fatty acid. As it elongates, the solubility in water decreases and consequently the melting point increases (greater consistency).
Fatty acids generally have an even number of carbon atoms, even if in some foods, such as vegetable oils, we find minimal percentages with odd numbers.
In the human body fatty acids are very abundant, but rarely free and mostly esterified with glycerol (triacylglycerols, glycerophospholipids) or with cholesterol (cholesterol esters).
Since each fatty acid is formed by an aliphatic (hydrophobic) carbon chain which

This characteristic heavily affects the entire digestive process of lipids.
Based on the presence or absence of one or more double bonds in the aliphatic chain, fatty acids are defined:
- saturated when their chemical structure does not contain double bonds,
- unsaturated when one or more double bonds are present
Cis and trans fatty acids

Based on the position of the hydrogen atoms associated with the carbons engaged in the double bond, a fatty acid can exist in nature in two forms, a cis and a trans.
The presence of a double bond in the aliphatic chain implies the existence of two conformations:
- cis if the two hydrogen atoms bonded to the carbons engaged in the double bond are arranged on the same plane
- trans if the spatial arrangement is opposite.
The cis form lowers the melting point of the fatty acid and increases its fluidity.

Two identical fatty acids, but which have a bond in the cis conformation and one in the trans conformation, have different names. The figure shows a fatty acid with eighteen carbon atoms, with unsaturation in position nine and cis conformation (oleic acid, the most abundant fatty acid in nature and present above all in olive oil); its trans isomer, present in very low percentages, takes on a different name (elaidinic acid).
Importance of the stereo-isomerism of the double bond

Let's look at the image; on the left a saturated fatty acid is represented, note the aliphatic chain (lipophilic tail) perfectly linear.
To its right we see the same fatty acid with a trans bond. The chain undergoes a small bending, but still remains a linear structure, similar to that of saturated fatty acid.
Further to the right we can appreciate the folding of the chain induced by the presence of a cis double bond. Finally, on the far right, the very strong folding associated with the presence of two unsaturated cis double bonds is represented.
This explains why butter, a food rich in saturated fatty acids, is solid at room temperature, while oils, in which cis unsaturated fatty acids prevail, are liquid under the same conditions. In other words, the presence of double cis bonds lowers the melting point of the lipid.
Where are trans fatty acids found?
To give greater consistency to the oils and unsaturated fats, processes (hydrogenation) have been devised in which the artificial breaking of a double bond and the hydrogenation of the product are carried out, thus obtaining foods in which the percentage of the trans form is high.
As already mentioned, natural unsaturated fats are normally found in the cis form. However, a small amount of trans fat is present in food, as it is formed in the stomach of ruminants due to the action of certain bacteria. For this reason, very small amounts of trans fatty acids are found in milk, dairy products and beef. The same are also found in the seeds and leaves of various plants, whose food consumption is however irrelevant.
The greatest health risks therefore derive from the massive use of hydrogenated oils and fats, which are abundant especially in margarines, sweet snacks and many spreads. This process takes place through the use of specific catalysts that subject the mixture of animal oils and fats to high temperatures and pressures, until chemically altered fatty acids are obtained. This process is particularly tempting to the food industries since it allows to obtain fats at a reduced cost. and with specific requirements (spreadability, compactness, etc.) Furthermore, the storage time is considerably extended, a fundamental aspect also from an economic point of view.
Why are trans fatty acids dangerous?
All this attention paid to trans fatty acids is due to the negative health implications that their use entails. These fatty acids in fact determine an increase in "bad cholesterol" (LDL lipoproteins) accompanied by a decrease in the "good" fraction (HDL lipoproteins). A high consumption of trans fatty acids, strongly represented in margarine and baked goods (snacks, spreads, etc.), therefore increases the risk of developing serious cardiovascular diseases (atherosclerosis, thrombosis, stroke, etc.).
What are NON-hydrogenated vegetable fats?
Today, the food industry is able to use alternative technologies to hydrogenation, to obtain vegetable fats free of dangerous trans fatty acids, but with the same organoleptic characteristics.
However, these are artificially manipulated products, not natural and perhaps made from poor quality or already rancid oils. Furthermore, they still have a high content of saturated fatty acids, precisely because they are semi-solid at room temperature.
Nomenclature of fatty acids
The nomenclature of fatty acids is very important, even if quite complex and in some respects controversial.
First of all it is necessary to quantify the length of the aliphatic chain, expressing it with the letter C followed by the number of carbons present in the fatty acid (eg C14, C16, C18, C20 etc.).
Secondly, it is necessary to indicate the number of unsaturations, following the symbol Cn with the symbol ":" followed by the number of double or triple bonds (for example, oleic acid, having a chain of 18 carbon atoms in which a only unsaturation, will be indicated by the abbreviation C18: 1).
Finally, it is necessary to specify where the possible unsaturation is found. In this regard, there are two different nomenclatures:
- the first refers to the position of the first unsaturated carbon that is encountered by starting to number the carbon chain from the initial carboxylic group; this position is indicated by the initials Δn, where n is, in fact, the number of carbon atoms present between the carboxyl end and the first double bond.
- In the second case, the numbering of the carbon atoms starts from the terminal methyl group (CH3); this position is indicated by the initials ωn, where n is, in fact, the number of carbon atoms present between the final methyl end and the first double bond
In the case of oleic acid the complete nomenclature is C18: 1 Δ9 or C18: 1 ω9.
The first numbering is preferred by food chemists, while in the medical field the second is preferred.
Examples:

Linoleic acid
C18: 2 Δ9.12 or C18: 2 ω6
Α-linolenic acid
C18: 3 Δ9,12,15 or C18: 3 ω3
Saturated fatty acids
With general formula CH3 (CH2) nCOOH they do not have double bonds and therefore cannot bond with any other element. The quantity of carbon atoms present in the aliphatic chain gives consistency to the substance, raising the melting point and changing its appearance at room temperature (solid). They are present both in fats of vegetable origin and in fats of animal origin, but they prevail clearly in the latter.
Main saturated fatty acids and their distribution in nature (From Chimica Degli Alimenti - Cabras, Martelli - Piccin)
Fusion point
(° C)
The fatty acids highlighted in bold are the most important from a nutritional point of view. The melting point is directly proportional to the number of carbon atoms present in the fatty acid; for this reason, foods rich in long-chain fatty acids have a greater consistency.

B.C. Lauricus (12: 0)
B.C. Myristic (14: 0)
B.C. Palmiticus (16: 0)
Saturated fatty acids and health
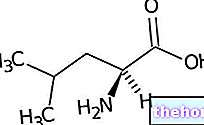
The saturated fatty acids in the diet raise cholesterol, so they are atherogenic. In this regard, it is useful to remember that saturated fatty acids do not all have the same atherogenic power. The most dangerous ones are palmitic (C16: 0), myristic (C14: 0) and lauric (C12: 0). Stearic (C18: 0), on the other hand, despite being saturated, is not very atherogenic, since the organism rapidly desaturates it, forming oleic acid.
Even medium-chain fatty acids are devoid of atherogenic power.
second part "



.jpg)


.jpg)