2,3 diphosphoglycerate (2,3 DPG) is a compound derived from an intermediate product of glycolysis; it is particularly concentrated at the erythrocyte level, since the red blood cells - being devoid of mitochondria - exploit the anaerobic lactacid metabolism (homolactic fermentation of glucose) to obtain energy.

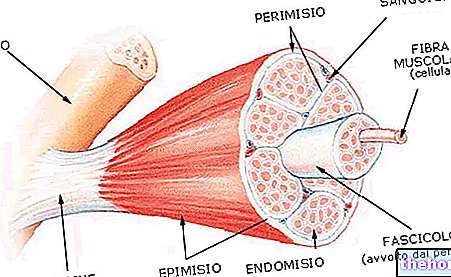
Hemoglobin is a tetrameric protein, which is made up of four subunits, two alpha and two beta, each made up of a protein portion (globin) and a heme (prosthetic group that binds oxygen). The 2,3-diphosphoglycerate attaches itself to the Beta chains, compacting them and reducing the affinity of hemoglobin for oxygen.
The binding of the 2,3 DPG to the hemoglobin occurs when it is in the deoxygenated form, while it is dissolved in the lung by the binding of the hemoglobin with the oxygen. In fact, when the hemoglobin reaches the tissues, the β chains are the first to releasing the oxygen and this loss involves a displacement of the monomers from the center. As soon as the internal hydrophilic cavity opens, the DPG enters and binds to the tetramer forming heteropolar bonds between its negatively charged groups and the lysine and histidine residues of the chains Beta, positively charged. The stabilized structure can also release the oxygen of the two α chains. In the lungs, however, the reverse process takes place; at high oxygen pressure the α chains are the first to bind it and the DPG is "squeezed" and expelled from the tetramer, allowing an easier Oxygen-β chain bond.
The 2,3 bisphosphoglycerate cannot bind to fetal hemoglobin, since this molecule is devoid of the B chains with which the 2,3 DPG binds. This explains the greater affinity for oxygen of fetal hemoglobin than maternal hemoglobin, characteristic which allows the fetal blood to extract oxygen from the maternal blood.





-cos-come-si-calcola-a-cosa-serve.jpg)