KONAKION ® is a drug based on Phytomenadione
THERAPEUTIC GROUP: Vitamin K and other antihemorrhagics

Indications KONAKION ® - Phytomenadione
KONAKION ® is used in the treatment of haemorrhagic diseases, both neonatal and induced by a deficiency in vitamin K dependent coagulation factors.
Mechanism of action KONAKION ® - Phytomenadione
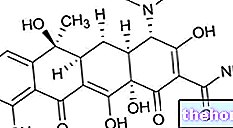
Phytomenadione, the active ingredient of KONAKION ®, also called phylloquinone, is a product of plant origin included in the vitamin K family, more precisely as vitamin K1.
This vitamin plays a crucial role in the activation of a series of coagulation factors (II, VII, IX and X), defined precisely as vitamin K-dependent factors, acting as a cofactor of a carboxylase responsible for the gamma carboxylation of a residue of glutamate exposed by the aforementioned factors.
This chemical reaction, which in addition to involving vitamin K also requires the intervention of epoxide reductase, takes the form of the biological activation of factors involved in the activation of coagulation both through the extrinsic and intrinsic pathway.
Alterations of this enzyme cascade are usually observed in patients undergoing long-term oral anticoagulant therapy and more rarely in malnourished newborns or in rare cases of congenital deficiency.
Fortunately, even oral therapy, thanks to the ability of vitamin K to be absorbed effectively by the ileal mucosa, allows the deficiencies present to be restored fairly quickly, thus being able to improve the bleeding diathesis.
Studies carried out and clinical efficacy
1. THE EFFECTS OF VITAMIN K1 ON TUMOR MARKERS
Dig Dis Sci. 2011 Jun; 56: 1876-83. Epub 2010 Dec 28.
Differential effects of vitamin K1 on AFP and DCP levels in patients with unresectable HCC and in HCC cell lines.
Carr BI, Wang Z, Wang M, Wei G.
Study that demonstrates how the administration of vitamin K1 can lead to a significant reduction in the blood concentrations of some tumor markers of hepatocarcinoma. These data could open up new experimental strategies.
2.SYNERGY BETWEEN VITAMIN K AND D
Altern Med Rev. 2010 Sep; 15: 199-222.
Vitamins D and K as pleiotropic nutrients: clinical importance to the skeletal and cardiovascular systems and preliminary evidence for synergy.
Kidd PM.
Very interesting work that emphasizes the pleiotropic and synergistic effects of the administration of vitamin D and K. Both vitamins have proved useful in the protection of skeletal and vascular structures, thus guaranteeing a preventive effect against chronic and degenerative pathologies.
3.VITAMIN K AND BONE HEALTH
Am J Clin Nutr. 2008 Aug; 88: 356-63.
Vitamin K, circulating cytokines, and bone mineral density in older men and women.
Shea MK, Dallal GE, Dawson-Hughes B, Ordovas JM, O "Donnell CJ, Gundberg CM, Peterson JW, Booth SL.
Work that demonstrates how low blood concentrations of vitamin K can be associated with an increased presence of inflammatory cytokines, involved in the bone demineralization process. Despite this observation, vitamin K supplementation was not effective in improving bone density parameters.
Method of use and dosage
KONAKION ®
Phytomenadione 10 mg chewable coated tablets;
Drops for oral solution of 20 mg of phytomenadione per ml of solution;
Oral and injectable solution of 10 mg phytomenadione per ml of solution;
Oral and injectable solution for infants of 2 mg of phytomenadione per 0.2 ml of solution.
While the 2 mg oral or injectable solution is indicated exclusively in the prophylaxis and treatment of neonatal haemorrhagic syndrome, associated with endogenous coagulation factor deficiencies, the other products are instead indicated in the treatment of hypoprothrombinemic haemorrhages.
The dosages to be used in the treatment of bleeding disorders should be defined by the physician, based on the results of coagulation tests and related symptoms.
All treatment should be supervised by a specialist doctor.
Warnings KONAKION ® - Phytomenadione
The administration of KONAKION ® should be preceded by a "careful medical examination useful to ascertain the absence of contextual pathologies, mainly atherosclerotic and thrombotic, potentially incompatible with the intake of antihemorrhagics.
In patients suffering from liver disease of any degree, the observed result in terms of bleeding control could significantly differ from that expected, therefore requiring a possible adjustment of the dosage used.
KONAKION ® in chewable tablets contains glucose and sucrose, therefore its intake is contraindicated in patients with diabetes, glucose-galactose malabsorption and fructose intolerance.
Furthermore, the presence of para-hydroxy-benzonates could increase the risk of allergic reactions.
PREGNANCY AND BREASTFEEDING
The intake of KONAKION ® during pregnancy and in the subsequent period of breastfeeding, should only be done in case of real need and under strict medical supervision.
Interactions
Given the biological role of phytomenadione, it is easy to imagine how taking KONAKION ® is able to completely antagonize the anticoagulant effects of dicumarins.
An adjustment of the vitamin K dosage should be expected following the possible concomitant intake of antibiotics and salicylates.
Contraindications KONAKION ® - Phytomenadione
KONAKION ® is contraindicated in case of hypersensitivity to the active ingredients or their excipients.
Undesirable Effects - Side Effects
The side effects described following the administration of KONAKION ® were observed mainly following the intravenous administration of the drug.
Bronchospasm, cyanosis, injection site pain, pruritus, tachycardia were the most clinically evident and recurrent adverse reactions among adults.
In children, on the other hand, jaundice, constipation, abdominal pain, skin rashes, and general malaise were observed.
Fortunately, in all cases, the symptoms returned with the simple suspension of medical therapy.
Note
KONAKION ® is a drug that can only be sold with a prescription.
The information on KONAKION ® - Fitomenadione published on this page may be out of date or incomplete. For a correct use of this information, see the Disclaimer and useful information page.