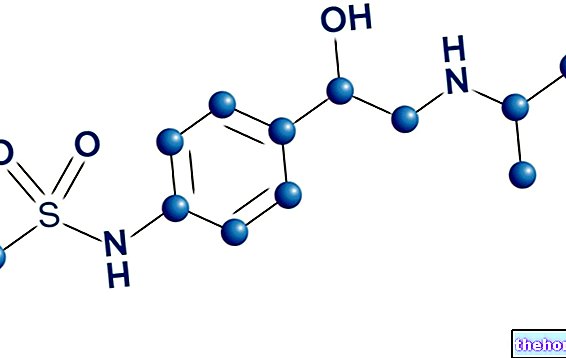
Active ingredients: Zolpidem (Zolpidem tartrate)
ZOLPIDEM SANDOZ 10 mg film-coated tablets
Why is Zolpidem used - Generic Drug? What is it for?
Zolpidem Sandoz is a hypnotic belonging to the group of benzodiazepine-like substances. It is indicated for the short-term treatment of sleep disorders.
Treatment with benzodiazepines and benzodiazepine-like substances is only prescribed for sleep disorders that are clinically relevant, disabling or causing extreme agitation.
Contraindications When Zolpidem - Generic Drug should not be used
Do not take Zolpidem Sandoz 10mg
- if you are allergic (hypersensitive) to zolpidem or any of the other ingredients of Zolpidem 10mg (see section 6)
- if you suffer from pathological muscle weakness (myasthenia gravis)
- if your breathing stops for short periods while you sleep (sleep apnea syndrome)
- if you suffer from severe respiratory weakness (where the lungs cannot get enough oxygen) (respiratory failure)
- if you suffer from severe liver damage (liver failure) because there is a risk of brain damage (encephalopathy).
Children and adolescents under the age of 18 should not take Zolpidem Sandoz.
Precautions for use What you need to know before taking Zolpidem - Generic Drug
General
Before starting treatment with Zolpidem Sandoz 10mg:
- the cause of sleep disturbances must be determined
- the underlying diseases must be treated.
If treatment for sleep disorders is unsuccessful after a period of 7 to 14 days, this may indicate a psychiatric or physical disorder to be evaluated. You should contact your doctor.
Effects observed after the use of benzodiazepines and benzodiazepine-like substances (such as Zolpidem Sandoz 10mg) or other hypnotics are as follows:
Dependence
The development of physical or psychological dependence is possible. This risk increases with dose and duration of treatment and is high in patients with a history of alcohol or drug abuse. In cases where physical dependence has developed, abrupt discontinuation of treatment is accompanied by withdrawal symptoms.
Memory problems (amnesia)
It can cause temporary memory loss (anterograde amnesia). This effect usually occurs several hours after taking Zolpidem Sandoz 10mg. To minimize this risk, you must be sure that you can enjoy an uninterrupted sleep of 8 hours (see section 4 "Possible side effects").
Psychiatric and "paradoxical" reactions
Restlessness, inner turmoil, irritability, aggression, delirium (psychosis), anger, nightmares, hallucinations, sleepwalking, inappropriate behavior, increased sleep disturbances and other behavioral side effects are known to occur during treatment. If this happens, you should stop taking Zolpidem Sandoz 10mg and contact your doctor. These reactions occur more easily in the elderly.
Some patients taking Zolpidem Sandoz 10mg who were not fully awake have been reported to have sleepwalking and other related behaviors, such as "driving in a state of sleep", preparing and eating food, making phone calls or having sexual intercourse, without have no recollection of the event. The risk of such events may increase if you take Zolpidem Sandoz 10mg with alcohol or other drugs that slow down the activity of the central nervous system, or if the maximum recommended dose is exceeded. If such events occur, tell your doctor immediately, who may advise you to stop treatment.
Psychomotor impairment in the next day (see also Driving and using machines)
The day after taking Zolpidem Sandoz 10mg, the risk of psychomotor impairment, including impaired ability to drive, may be increased if you:
- take this medicine less than 8 hours before performing activities that require mental alertness
- take a higher dose than recommended
- take zolpidem while already taking other central nervous system depressant drugs or other medicines that increase zolpidem blood levels, or while drinking alcohol, or while taking illicit drugs Take the single dose immediately at bedtime. Do not take another dose on the same night.
Benzodiazepines and benzodiazepine-like substances (such as Zolpidem Sandoz 10mg) should not be used as the only treatment for delirium (psychosis) and depression or anxiety accompanied by depression.
Special patient groups
- If you are an elderly and debilitated patient. You should be given a lower dosage (see section 3. "How to take Zolpidem Sandoz 10mg"). Zolpidem Sandoz 10mg exerts a muscle relaxant effect. For this reason, older people in particular are at risk of falling and causing hip fractures when they get out of bed at night.
- If you have impaired kidney function. Your body will need more time to get rid of Zolpidem Sandoz 10mg. Although no dosage adjustment is necessary, caution is warranted. Talk to your doctor.
- If you have chronic dyspnea problems. Your breathing problems can get worse.
- If you have a history of alcohol and drug abuse. These patients should be carefully monitored during treatment with Zolpidem Sandoz 10mg as there is a risk of addiction and psychological dependence.
- If you have severe liver problems. You should not use Zolpidem Sandoz 10mg if you are at risk of brain damage (encephalopathy). Talk to your doctor.
- If you suffer from delirium (psychosis), depression or anxiety related to depression, Zolpidem Sandoz 10mg should not be the only treatment you receive.
Interactions Which drugs or foods may change the effect of Zolpidem - Generic Drug
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines including medicines obtained without a prescription.
Some medicines may be affected by the effects of Zolpidem Sandoz 10mg. These medicines, in turn, can affect the effectiveness of Zolpidem Sandoz. You may experience more sleepiness when you take Zolpidem Sandoz 10mg in combination with the medicines listed below.
Zolpidem Sandoz 10mg can interact with:
- strong painkillers (analgesics-narcotics). An increased feeling of well-being (euphoria) may occur which can lead to an increase in psychological dependence
- muscle relaxants
- medicines used to treat a variety of fungal infections, such as itraconazole and ketoconazole. Taking ketoconazole with Zolpidem Sandoz 10mg may increase the sleepiness effect
- clarithromycin and erythromycin (antibiotics)
- ritonavir (an anti-retroviral drug used to treat HIV and AIDS).
When taking zolpidem with the following medicines, increased effects of somnolence and psychomotor impairment in the next day, including impaired ability to drive, are possible.
- medicines for certain mental health problems (antipsychotics)
- medicines for insomnia problems (hypnotics)
- medicines to calm or reduce anxiety
- medicines for depression
- medicines for moderate to severe pain (narcotic analgesics)
- medicines for epilepsy
- medicines used for anesthesia
- medicines for hay fever, skin rash (rash) or other allergies, which can make you sleepy (sedative antihistamines)
While you are taking zolpidem with antidepressant medicines, including bupropion, desipramine, fluoxetine, sertraline and venlafaxine, you may see non-existent things (hallucinations). It is not recommended to take zolpidem with fluvoxamine or ciprofloxacin.
Medicines that strongly enhance the activity of some liver enzymes, such as rifampicin (an antibacterial used to treat, for example, tuberculosis), may reduce the effect of Zolpidem Sandoz 10mg.
Taking Zolpidem Sandoz 10mg with food and drink
You should not consume alcohol during treatment, as the effect of zolpidem may be increased, making you more sleepy or sluggish. Your ability to perform tasks that require increased concentration, such as driving or operating machinery will be impaired.
Warnings It is important to know that:
Pregnancy and breastfeeding
Tell your doctor if you want to become pregnant or if you suspect that you are pregnant during treatment with Zolpidem Sandoz 10mg, so that your doctor can decide whether to continue the treatment or not.
Zolpidem Sandoz 10mg should not be taken during pregnancy and breastfeeding, especially in the first three months as there is insufficient data available to ascertain the safe use of Zolpidem Sandoz 10mg during pregnancy and breastfeeding.
However, if the benefit to the mother outweighs the risk to the baby, the doctor may decide to treat her with Zolpidem Sandoz 10mg. If Zolpidem Sandoz 10mg is taken for a longer period during the last months of pregnancy, withdrawal symptoms may occur after birth in the baby.
Although zolpidem is excreted in breast milk in small amounts, Zolpidem Sandoz 10mg should not be taken while breastfeeding.
Ask your doctor or pharmacist for advice before taking any medicine.
Driving and using machines
Zolpidem Sandoz 10mg impairs the ability to drive and use machines, for example with the risk of 'falling asleep at the wheel'.
On the day after taking Zolpidem Sandoz 10mg (as with other hypnotic medicines), you should be aware that:
- you may feel drowsy, sleepy, dizzy or confused
- it may take longer to make decisions
- vision may be blurred or double
- may feel less alert
A period of at least 8 hours is recommended between taking zolpidem and driving vehicles, using machines and working at height to minimize the effects listed above.
Do not drink alcohol or take other psychoactive substances while taking Zolpidem Sandoz 10mg, as the effects listed above may be enhanced.
Important information about some of the ingredients of Zolpidem Sandoz 10mg
Zolpidem Sandoz 10mg contains lactose. If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this medicinal product.
Dose, Method and Time of Administration How to use Zolpidem - Generic Drug: Posology
Always take Zolpidem Sandoz 10mg exactly as your doctor has told you. If in doubt, consult your doctor or pharmacist.
The tablets should be taken with liquid immediately before going to sleep.
The usual dose is:
Adults
The recommended dose every 24 hours is 10mg of Zolpidem Sandoz 10mg. A lower dose may be prescribed for some patients. Zolpidem Sandoz 10mg should be taken:
- with a single administration,
- just before bedtime.
Make sure you have a period of at least 8 hours after taking this medicine before performing any activities that require your vigilance.
Do not exceed 10 mg every 24 hours.
Elderly and debilitated patients and patients with hepatic insufficiency:
The recommended dose is 1/2 tablet of Zolpidem Sandoz 10 mg (equivalent to 5 mg).
This dose should only be increased to 1 tablet of Zolpidem Sandoz 10mg (equivalent to 10mg) if the effect is insufficient and if the medicine is well tolerated.
Children and adolescents
Zolpidem Sandoz 10mg should not be given to children and adolescents under 18 years of age.
How long should Zolpidem Sandoz 10mg be taken?
After repeated intake over the course of several weeks, the hypnotic effect (which promotes sleep) may be reduced.
The duration of treatment should be as short as possible. In general, the duration of treatment varies from a few days to 2 weeks and should not exceed 4 weeks including the period of gradual dose reduction. Your doctor will tell you how to gradually reduce the dosage to avoid withdrawal effects.
In special cases, it may be necessary to extend the treatment beyond 4 weeks. Your doctor will evaluate your response to treatment and decide if this is necessary.
If you forget to take Zolpidem Sandoz 10mg
Do not take a double dose to make up for a forgotten dose.
If you forget to take a dose, you can take it as long as you can still sleep for 7 or 8 hours. If this is not possible, you should not take the tablet until you go to sleep the next day.
Then continue to take Zolpidem Sandoz 10mg as prescribed by your doctor.
If you stop taking Zolpidem Sandoz 10mg
Do not suddenly stop taking Zolpidem Sandoz 10mg. The risk of onset of withdrawal symptoms (see section 2 under "withdrawal symptoms") is higher after abrupt discontinuation of treatment. Your doctor will advise you on how to end the treatment by gradually reducing the dose.
If the treatment is stopped suddenly, the quality of sleep can temporarily deteriorate (rebound phenomenon). This may be accompanied by mood changes, anxiety and agitation. Withdrawal symptoms consist of headache or muscle pain, extreme anxiety and tension, restlessness, confusion, irritability and sleep disturbances and in severe cases loss of perception may occur. reality (derealization), alienation from oneself (depersonalization), abnormal sensitivity to sounds (hyperacusis), numbness and tingling in the extremities, hypersensitivity to light, noise and physical contact, hallucinations or convulsions (seizures).
It is important to be aware of the possibility of such symptoms occurring so that you are able to minimize anxiety.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
Overdose What to do if you have taken too much Zolpidem - Generic Drug
In case of overdose contact your doctor immediately. Take any remaining tablets in the carton or package leaflet with you so that staff know exactly what you have taken.
In the event of an overdose, symptoms can range from extreme sleepiness to mild coma, possibly to fatal coma.
Side Effects What are the side effects of Zolpidem - Generic Drug
Like all medicines, Zolpidem Sandoz 10mg can cause side effects, although not everybody gets them.
These effects appear to be related to personal sensitivity and appear more frequently in the hours after taking the tablet if you do not go to bed or sleep immediately.
These side effects occur more commonly in elderly patients.
The side effects are as follows:
- Very common: affects more than 1 in 10 patients
- Common: affects 1 to 10 users in 100
- Uncommon: affects 1 to 10 users in 1000
- Rare: affects 1 to 10 users in 10,000
- Very rare: affects less than 1 in 10,000 patients.
- Not known: frequency cannot be estimated from the available data
Common (affects 1 to 10 users in 100):
- Mental: feeling things that are not real (hallucinations), agitation, nightmares
- Nervousness: sleepiness during the next day, emotional numbness, decreased attention, fatigue, headache, dizziness, difficulty remembering things, which may be associated with inappropriate behavior, ataxia (loss of muscle coordination), worsening of insomnia
- Auditory: spinning sensation with loss of balance (vertigo)
- Gastric and intestinal: diarrhea, nausea, vomiting, stomach pain
- General Disorders and Administration Site Conditions: Fatigue
Uncommon (affects 1 to 10 users in 1000):
- Mental: state of confusion, irritability
- Eyepieces: double vision
Frequency not known (frequency cannot be estimated from the available data):
- Mental: restlessness, aggression, delirium, anger, mental disorders (psychosis), sleepwalking, inappropriate behavior and other negative behavioral effects, memory loss (amnesia), which may be associated with improper behavior (see section 2 "Warnings and precautions ").
Such reactions are more likely in the elderly.
Pre-existing depression may become apparent when using Zolpidem Sandoz 10mg or other sleep-promoting drugs (hypnotics).
Taking Zolpidem Sandoz 10 mg over a long period of time may cause physical or psychological dependence. If you suddenly stop taking Zolpidem Sandoz 10 mg, you may experience withdrawal symptoms (see section 2, "Warnings and precautions"). .
Misuse of Zolpidem Sandoz 10 mg by drug addicts has been reported
- Decreased sexual need (libido)
- Cutaneous: rash, hives, itching, excessive sweating
- Muscles: muscle weakness
- General disorders and administration site conditions: abnormal posture when walking (gait disturbances), drug tolerance, falls (especially in elderly patients and when Zolpidem 10 mg was not taken as prescribed)
- Hepatic: increase in liver enzymes
- Immune system: Sudden swelling of the lips, cheeks, eyelids, tongue or throat.
If any of these occur, you should stop taking Zolpidem Sandoz 10mg and contact your doctor.
Pre-existing depression may become manifest during the use of benzodiazepines or benzodiazepine-like substances.
Taking Zolpidem Sandoz 10mg (even at therapeutic dosages) can lead to physical dependence: interruption of therapy can cause suspension or rebound phenomena (see "Warnings and precautions").
Psychological dependence can arise. Misuse of Zolpidem Sandoz 10mg has been reported in drug abusers.
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet please tell your doctor or pharmacist.
Expiry and Retention
Keep Zolpidem Sandoz 10mg out of the reach and sight of children.
Store in the original container to protect from light.
Do not use Zolpidem Sandoz 10mg after the expiry date which is stated on the carton and blister.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. This will help protect the environment.
Deadline "> Other information
What Zolpidem Sandoz 10mg contains
The active ingredient is zolpidem tartrate 10 mg. Each tablet contains 10 mg of zolpidem tartrate.
The other ingredients are:
- Tablet core: succinic acid, sodium carboxymethyl starch (type A), microcrystalline cellulose, lactose monohydrate, magnesium stearate, colloidal silicon dioxide.
- Tablet coating: lactose monohydrate, macrogol 4000, hypromellose, coloring agent titanium dioxide (E171).
What Zolpidem Sandoz 10mg looks like and contents of the pack
Zolpidem Sandoz 10mg are white, oblong, biconvex film-coated tablets with breakline.
The film-coated tablets are contained in blister packs of: 10, 20, 28, 30, 30x1, 50, 98, 100 film-coated tablets.
Not all pack sizes may be marketed.
Source Package Leaflet: AIFA (Italian Medicines Agency). Content published in January 2016. The information present may not be up-to-date.
To have access to the most up-to-date version, it is advisable to access the AIFA (Italian Medicines Agency) website. Disclaimer and useful information.
01.0 NAME OF THE MEDICINAL PRODUCT -
ZOLPIDEM SANDOZ 10 MG TABLETS COATED WITH FILM
02.0 QUALITATIVE AND QUANTITATIVE COMPOSITION -
One film-coated tablet contains:
10 mg of zolpidem tartrate
Excipients: 54 mg lactose / film-coated tablet
For the full list of excipients see section 6.1
03.0 PHARMACEUTICAL FORM -
Film-coated tablet.
White, oblong, biconvex, with fracture line. The tablet can be divided into equal halves.
04.0 CLINICAL INFORMATION -
04.1 Therapeutic indications -
Short-term treatment of insomnia.
Benzodiazepines or benzodiazepine-like substances are only indicated when the disorder is severe, debilitating or likely to cause profound discomfort.
04.2 Posology and method of administration -
Treatment should be as short as possible. Generally, the duration of treatment varies from a few days to 2 weeks, up to a maximum of 4 weeks, including the phase of gradual treatment decrease. The period of tapering off treatment should be tailored to individual needs.
In particular cases, an extension beyond the maximum treatment period may be necessary; in these cases the extension should not be carried out without re-evaluating the patient's status.
Take the product with liquid before going to bed.
Adults:
The treatment should be taken as a single administration and should not be re-administered during the same night.
The recommended daily dose is 10 mg, to be taken immediately at bedtime. The total daily dose of zolpidem should not exceed 10 mg.
Elderly patients
For elderly or debilitated patients who may be particularly sensitive to the effects of zolpidem, the recommended dose is 5 mg. This dose should only be increased to 10 mg when the clinical response is inadequate and the drug is well tolerated. The total dose of 10 mg should not be exceeded in any patient.
Patients with hepatic insufficiency
For patients with hepatic insufficiency who do not clear the drug as quickly as healthy subjects, the recommended dose is 5 mg. This dose should only be increased to 10 mg when the clinical response is inadequate and the drug is well tolerated.
The total dose of 10 mg should not be exceeded in any patient.
Children and adolescents under 18 years of age:
Zolpidem is contraindicated in children and adolescents under 18 years of age.
04.3 Contraindications -
Severe hepatic insufficiency
Hypersensitivity to zolpidem or to any of the excipients
Sleep Apnea Syndrome
Myasthenia gravis
Severe respiratory failure
Children and adolescents under 18 years of age
04.4 Special warnings and appropriate precautions for use -
General
When possible, determine the cause of the insomnia. Before prescribing a hypnotic, treat the underlying conditions. Failure to treat insomnia after a period of 7-14 days may indicate the presence of a primary psychiatric disorder or a physical ailment to be evaluated.
General information regarding the effects observed after administration of benzodiazepines or other hypnotic substances which must be taken into consideration by the prescribing physician is described below.
Tolerance
After repeated use for a few weeks, some reduction in the hypnotic effect of short-lived benzodiazepines or other benzodiazepine-like substances may occur.
Dependence
The use of benzodiazepines or other benzodiazepine-like substances may induce the development of physical or psychological dependence on these products. The risk of dependence increases with dose and duration of treatment and is also greater in patients with a history of the disorder. psychiatric and / or alcohol or drug abuse These patients should be monitored carefully while receiving benzodiazepines or benzodiazepine-like substances.
In cases where physical dependence has developed, abrupt discontinuation of treatment will be characterized by the manifestation of withdrawal symptoms. These can include headaches or muscle pain, extreme anxiety and tension, restlessness, confusion and irritability. In severe cases, the following symptoms are possible: derealization, depersonalization, hyperacusis, numbness and tingling in the extremities, hypersensitivity to light, noise and physical contact, hallucinations or seizures.
Return / rebound insomnia
Upon discontinuation of the hypnotic drug, a transient syndrome may occur which consists in the reappearance, in an accentuated form, of the symptoms that led to treatment with a benzodiazepine or with a substance similar to benzodiazepines. This syndrome can be accompanied by other types of reactions such as mood swings, anxiety and agitation.
It is important that the patient is informed of the possible occurrence of return / rebound phenomena, so that when these symptoms occur after the drug has been discontinued, the resulting anxiety is reduced to a minimum.
It seems that, in the case of benzodiazepines and other short-acting benzodiazepine-like substances, withdrawal phenomena may occur in the interval between two intakes, especially when the dose is high.
As the risk of withdrawal symptoms / return / rebound phenomena is more likely to occur after abrupt discontinuation of treatment, tapering of the dose is recommended.
Duration of treatment
The duration of treatment should be as short as possible (see section 4.2), but should not exceed 4 weeks including the tapering off phase. An extension of this period should not occur without a reassessment of the patient's status.
It may be useful to inform the patient, when treatment is started, that it will be of limited duration.
Psychomotor impairment on the following day
The risk of psychomotor impairment in the next day, including impaired ability to drive, increases if:
• zolpidem is taken when less than 8 hours remain before performing activities that require mental alertness (see section 4.7);
• a higher dose than recommended is taken;
• zolpidem is co-administered with other central nervous system (CNS) depressant drugs, or other drugs that increase the blood levels of zolpidem, or with alcohol or illicit drugs (see section 4.5).
Zolpidem should be taken as a single administration, immediately at bedtime, and should not be re-administered during the same night.
Amnesia
Benzodiazepines and other benzodiazepine-like substances can cause "anterograde amnesia. This effect usually occurs several hours after taking the drug. To reduce the risk, patients should ensure that they can sleep continuously for 8 hours (see section 4.8). ).
Psychiatric and "paradoxical" reactions
When using benzodiazepines or other benzodiazepine-like substances, reactions such as restlessness, agitation, irritability, aggression, delusions, anger, nightmares, hallucinations, psychosis, sleepwalking, inappropriate behavior, increased insomnia and other side effects of known behavioral type. Should this occur, use of the drug should be discontinued. These reactions occur more easily in the elderly.
Particular groups of patients
• Elderly or debilitated patients
They should take a lower dose: see the recommended dose
(Paragraph 4.2).
Because of the "muscle relaxant effect" there is a risk of falls and consequent hip fractures, especially for elderly patients when they get up during the night.
• Patients with renal insufficiency (see section 5.2)
Although a different dosage is not necessary, use with caution.
• Patients with chronic respiratory insufficiency
Caution should be given to prescribing zolpidem as benzodiazepines have been shown to impair the respiratory center. It should also be borne in mind that anxiety and agitation are described as signs of decompensated respiratory failure.
• Patients with severe hepatic insufficiency
Benzodiazepines and benzodiazepine-like substances are not indicated for the treatment of patients with severe hepatic insufficiency as they may worsen the encephalopathy.
• Use in patients with psychotic disorders
Benzodiazepines and benzodiazepine-like substances are not recommended for primary treatment.
• Use in depression
Although important clinical, pharmacokinetic and pharmacodynamic interactions with SSRIs have not been demonstrated, zolpidem should be administered with caution to patients presenting with depressive symptoms. Suicidal tendencies may be present. Given the possibility of intentional overdose by the patient, dispense the lowest possible amount of the drug to these patients.
Benzodiazepines and benzodiazepine-like substances should not be used as the only treatment for depression or anxiety associated with depression (they can lead to suicide in this type of patient).
Latent depression may occur during use of zolpidem. Since insomnia may be a symptom of depression, the patient should be reevaluated if insomnia persists.
• Use in patients with a history of alcohol or drug abuse Benzodiazepines and benzodiazepine-like substances should be used with extreme caution in patients with a history of alcohol or drug abuse. These patients should be closely monitored when receiving zolpidem as they are at risk of addiction and psychic dependence.
Sleepwalking and associated behaviors
In some patients taking zolpidem who were not fully awake, there have been reports of sleepwalking and other associated behaviors such as "driving while asleep", preparing and eating food, making phone calls or having sexual intercourse, without having any recollection of "sleep". The risk of such events may increase if zolpidem is taken with alcohol or other drugs that slow down the activity of the central nervous system, or if the maximum recommended dose is exceeded. Discontinuation of zolpidem should be strongly considered in patients reporting such behaviors (see sections 4.5 and 4.8).
Zolpidem Sandoz 10 mg contains lactose. Patients with rare hereditary problems of galactose intolerance, the Lapp-lactase deficiency or glucose-galactose malabsorption should not take this medicine.
04.5 Interactions with other medicinal products and other forms of interaction -
Concomitant use with alcohol is not recommended. The sedative effect may increase if the drug is taken in combination with alcohol. This affects the ability to drive and use machines.
Association with CNS depressant drugs
An enhancement of the central depressive effect may occur in cases of concomitant use with antipsychotics (neuroleptics), hypnotics, anxiolytics / sedatives, antidepressants, narcotic analgesics, antiepileptic drugs, anesthetics and sedative antihistamines. Therefore, the concomitant use of zolpidem with such drugs. may increase somnolence and psychomotor impairment in the next day, including impaired ability to drive (see section 4.4 and section 4.7). In addition, there have been isolated reports of visual hallucinations in patients taking zolpidem with antidepressants, including bupropion, desipramine, fluoxetine, sertraline and venlafaxine.
Co-administration of fluvoxamine may increase blood levels of zolpidem; concomitant use is not recommended.
In the case of narcotic analgesics, an increase in euphoria is also possible, which can lead to an enhancement of psychological dependence.
CYP450 inhibitors and inducers Zolpidem is metabolised by certain enzymes of the cytochrome P450 family. The main enzyme is CYP3A4 with partial contribution of CYP1A2.
Rifampicin induces the metabolism of zolpidem; this results in a reduction in peak plasma concentration of approximately 60% and a possible decrease in efficacy. Similar effects can also be seen with other strong inducers of cytochrome P450 enzymes.
Substances that inhibit liver enzymes (particularly CYP3A4) may increase plasma concentration and increase the activity of zolpidem. However, when zolpidem is administered with itraconazole (inhibitor of CYP3A4), the pharmacokinetic and pharmacodynamic effects are not are significantly different. The clinical relevance of these findings is unknown.
Co-administration of ciprofloxacin may increase blood levels of zolpidem; concomitant use is not recommended.
Concomitant administration of zolpidem and ketoconazole, which is a potent inhibitor of CYP3A4, prolonged the half-life of zolpidem. Total exposure of zolpidem increased by 83% and apparent oral clearance decreased. No dose adjustment is required routinely, but the patient should be informed of the potential for increased sedative effect when ketoconazole and zolpidem are co-administered. . A dose reduction of zolpidem may be considered when initiating treatment with ketoconazole.
There is clinical experience that sertraline, a CYP3A4 inhibitor, may interact with zolpidem by increasing somnolence. Additionally, isolated cases of visual hallucinations have been reported.
No significant pharmacokinetic interactions were observed when zolpidem was administered with warfarin, haloperidol, chlorpromazine, digoxin or ranitidine.
04.6 Pregnancy and breastfeeding -
There are insufficient data on Zolpidem Sandoz to assess its safe use during pregnancy and lactation. Although animal studies have shown no teratogenic or embryotoxic effects, safety during pregnancy has not been confirmed in humans. Therefore zolpidem should not be used during pregnancy, especially during the first trimester.
If the product is prescribed to a woman of childbearing age, the patient should be advised of the need to contact her doctor to stop treatment in the event of a planned or suspected pregnancy.
If, for urgent medical needs, zolpidem is administered during the last phase of pregnancy, or during labor, effects on the newborn may occur, such as hypothermia, hypotonia and moderate respiratory depression, due to the pharmacological effect of the drug.
Discontinuation symptoms due to
development of physical dependence.
Zolpidem is excreted in breast milk in minimal quantities. Therefore, zolpidem should not be used during breastfeeding until the effects on the newborn are studied.
04.7 Effects on ability to drive and use machines -
Zolpidem Sandoz impairs the ability to drive and use machines.
Vehicle drivers and machine operators should be advised that, as with other hypnotics, there is a possible risk of drowsiness, prolonged reaction time, dizziness, drowsiness, confused / double vision and reduced alertness and impaired ability to drive, the morning after therapy (see section 4.8). To minimize the risk, a rest period of at least 8 hours is recommended between taking zolpidem and driving a vehicle, using machinery and working at height.
Impaired ability to drive and behaviors such as "falling asleep at the wheel" have occurred with zolpidem alone, at therapeutic doses.
Furthermore, co-administration of zolpidem with alcohol and other CNS depressant drugs increases the risk of such behaviors (see sections 4.4 and 4.5). Patients should be advised not to use alcohol or other psychoactive substances while taking zolpidem.
04.8 Undesirable effects -
The following frequency data are the basis for the evaluation of undesirable effects:
Very common (≥ 1/10) Common (≥ 1/100 to
Uncommon (≥ 1 / 1,000 to
Not known (frequency cannot be estimated from the available data)
There is evidence of a dose relationship for undesirable effects associated with the use of zolpidem tartrate, particularly for certain CNS and gastrointestinal events.
These side effects occur more commonly in elderly patients. These effects appear to be related to individual sensitivity and are most often seen during the hours following drug intake if the patient does not go to sleep or does not fall asleep immediately (see section 4.2).
Disorders of the immune system
Not known: angioneurotic edema
Psychiatric disorders
Common: hallucinations, agitation, nightmares
Uncommon: confusional state, irritability
Not known: restlessness, aggression, delusions, anger, psychosis, sleepwalking (see section 4.4), inappropriate behavior and other behavioral undesirable effects (these reactions occur more easily in the elderly, see section 4.4).
Pre-existing depression may become manifest during the use of benzodiazepines or substances similar to benzodiazepines (see section 4.4). Use (even at therapeutic doses) can induce physical dependence: discontinuation of therapy can cause withdrawal or withdrawal phenomena. return / rebound (see section 4.4).
Psychological dependence can arise. Abuse has been reported in poly-drug addicts.
Loss of libido.
Nervous system disorders
Communand: somnolence, somnolence during the next day, lightheadedness, reduced alertness, headache, dizziness, anterograde amnesia which may be associated with inappropriate behavior, ataxia, exacerbated insomnia.
Not known: reduced level of consciousness
Eye disorders
Uncommon: double vision.
Ear and labyrinth disorders
common: dizziness, ataxia.
Gastrointestinal disorders
common: gastrointestinal disturbances (diarrhea, nausea, vomiting, abdominal pain)
Hepatobiliary disorders
Not known: increased liver enzymes
Skin and subcutaneous tissue disorders
Uncommon: rash, itching, urticaria, hyperhidrosis, angioneurotic edema (Quincke's edema)
Musculoskeletal and connective tissue disorders
Not known: muscle weakness
General disorders and administration site conditions
Common: fatigue
Not known: difficulty walking, drug tolerance, falls (especially in elderly patients and when zolpidem tartrate has not been taken as prescribed)
04.9 Overdose -
In cases of overdose of zolpidem alone, impaired consciousness ranges from somnolence to alert coma and several severe symptoms including fatal cases.
After overdoses of up to 400 mg, ie 40 times the recommended dose, recovery was complete.
Establish general symptomatic and supportive treatment. If deemed appropriate, perform a gastric lavage immediately. Administer intravenous fluids if needed. If gastric emptying offers no benefit, administer activated charcoal to reduce absorption. Control of respiratory and cardiovascular functions should be considered. Do not administer sedatives, even if agitated.
Consider using flumazenil if severe symptoms are observed. Administration of flumazenil may contribute to neurological symptoms (convulsions). When treating overdose with any pharmaceutical product, be aware that multiple substances may have been taken by the patient.
Given the high volume of distribution and high protein binding capacity of zolpidem, hemodialysis and induced diuresis are not effective measures. Hemodialysis studies in patients with renal insufficiency treated with therapeutic doses have shown that zolpidem is not dialysable.
05.0 PHARMACOLOGICAL PROPERTIES -
05.1 "Pharmacodynamic properties -
Pharmacotherapeutic group: Hypnotics and Sedatives, Benzodiazepine analogues
ATC code: N05CF02
Zolpidem, an imidazopyridine, is a hypnotic substance similar to benzodiazepines. Experimental studies have shown sedative effects at doses lower than those necessary to obtain anticonvulsant, muscle relaxant or anxiolytic effects. These effects are related to a specific agonist action on the central receptors belonging to the macromolecular GABA-omega receptor complex (BZ1 and BZ2), which modulates the opening of the chlorine ion channels. Zolpidem acts primarily on the omega receptor subtypes (BZ1). The clinical significance of this finding is unknown.
Randomized trials have shown only convincing evidence of the efficacy of zolpidem 10 mg.
In a randomized double-blind study of 462 healthy volunteers not in old age suffering from transient insomnia, zolpidem 10 mg reduced the mean time to fall asleep by 10 minutes compared to placebo, while in the case of 5 mg zolpidem this time was 10 minutes. 3 minutes.
In a randomized double-blind study of 114 non-elderly patients suffering from chronic insomnia, zolpidem 10 mg reduced the mean time to fall asleep by 30 minutes compared to placebo, while in the case of 5 mg zolpidem this time was 15. minutes.
In some patients, a lower dose of 5 mg may be effective.
05.2 "Pharmacokinetic properties -
Absorption
Both absorption and onset of the hypnotic effect of zolpidem are rapid. Bioavailability after oral administration is 70%. Within the therapeutic dose range, the kinetics are linear. The therapeutic plasma level is between 80 and 200 ng / ml. The peak plasma concentration is reached between 30 minutes and 3 hours after administration.
The interindividual variability is high, (CV% of the AUC is 60-70% and for the
Cmax of 40-50%).
Distribution
The volume of distribution in adults is 0.54 L / kg and in the elderly it is reduced to 0.34 L / kg.
Plasma protein binding is 92%. First pass metabolism through the liver is about 35%. Protein binding is not affected by repeated administration, indicating the lack of a competitive effect between zolpidem and its metabolites for binding sites.
Elimination
The elimination half-life is short, with an average of 2.4 hours and a duration of action of up to 6 hours.
All metabolites are pharmacologically inactive and are excreted in the urine (56%) and faeces (37%).
Clinical studies have shown that zolpidem is not dialysable. Clearance is approximately 300 ml / min.
Particular groups of patients
A moderate reduction in clearance was observed in patients with renal insufficiency (independent of dialysis). The other pharmacokinetic parameters remain unchanged.
In elderly patients and in patients with hepatic insufficiency, the bioavailability of zolpidem is increased.
Clearance is reduced to approximately 100 ml / min in the elderly.
The maximum plasma concentration increased by approximately 80% without a significant increase in the half-life (approximately 3 hours) in patients aged 81 to 95 years.
05.3 Preclinical safety data -
Preclinical effects have only been observed at doses well above the maximum human exposure levels and are therefore of little importance for clinical use.
06.0 PHARMACEUTICAL INFORMATION -
06.1 Excipients -
Core of the tablet:
Magnesium stearate
Microcrystalline cellulose
Lactose monohydrate
Colloidal silicon dioxide
Sodium carboxymethyl starch (Type A)
Succinic acid
Coating:
Lactose monohydrate
Macrogol 4000
Hypromellose
Titanium dioxide (color E 171)
06.2 Incompatibility "-
Not relevant
06.3 Period of validity "-
5 years
06.4 Special precautions for storage -
Store in the original package to protect from light.
06.5 Nature of the immediate packaging and contents of the package -
The film-coated tablets are packed in PVC / aluminum blisters placed in a cardboard box.
Packs contain 10, 20, 28, 30, 30 x 1, 50, 98 and 100 film-coated tablets.
Not all pack sizes may be marketed.
06.6 Instructions for use and handling -
No special instructions
07.0 HOLDER OF THE "MARKETING AUTHORIZATION" -
Sandoz S.p.A. Largo U.Boccioni, 1 21040 Origgio (VA)
08.0 MARKETING AUTHORIZATION NUMBER -
10 mg film-coated tablets 10 tablets in PVC / AL blister AIC No. 038001018
10 mg film-coated tablets 20 tablets in PVC / AL blister AIC No. 038001020
10 mg film-coated tablets 28 tablets in PVC / AL blister AIC No. 038001032
10 mg film-coated tablets 30 tablets in PVC / AL blister AIC No. 038001044
10 mg film-coated tablets 50 tablets in PVC / AL blister AIC No. 038001057
10 mg film-coated tablets 98 tablets in PVC / AL blister AIC No. 038001069
10 mg film-coated tablets 100 tablets in PVC / AL blister AIC No. 038001071
10 mg film-coated tablets 30X1 tablets in PVC / AL AIC blister No. 038001083
09.0 DATE OF FIRST AUTHORIZATION OR RENEWAL OF THE AUTHORIZATION -
January 16, 2008