Active ingredients: hyoscine N-butylbromide
BUSCOPAN 10 mg coated tablets
BUSCOPAN 10 mg suppositories
Buscopan package inserts are available for pack sizes: - BUSCOPAN 10 mg coated tablets, BUSCOPAN 10 mg suppositories
- BUSCOPAN 20 mg / ml solution for injection
Why is Buscopan used? What is it for?
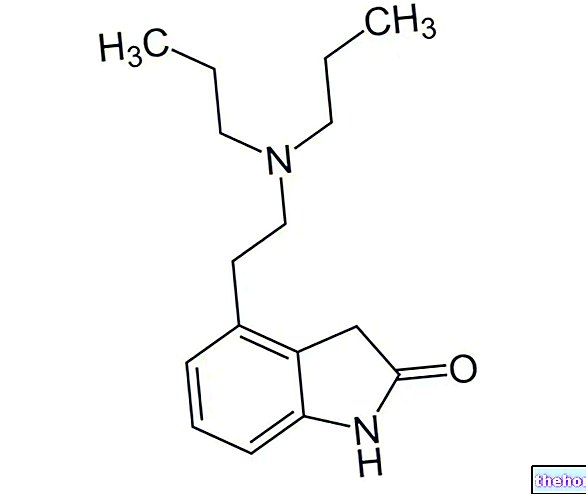
Buscopan is an antispasmodic, anticholinergic, belonging to the class of semisynthetic nightshade alkaloids, quaternary ammonium compounds.
Buscopan is used in the symptomatic treatment of spastic and painful manifestations of the gastrointestinal and genitourinary tract.
Contraindications When Buscopan should not be used
- Hypersensitivity to the active substance or to any of the excipients.
- Acute angle glaucoma.
- Prostatic hypertrophy or other causes of urinary retention.
- Pyloric stenosis and other conditions stenosing the gastrointestinal canal.
- Paralytic ileus, ulcerative colitis, megacolon.
- Reflux esophagitis.
- Intestinal atony of the elderly and debilitated subjects.
- Myasthenia gravis.
- Children under the age of 6.
In case of rare hereditary conditions of incompatibility with one of the excipients, the use of the medicinal product is contraindicated.
Precautions for use What you need to know before taking Buscopan
If you have severe abdominal pain of which the cause is not known, which persists or worsens, or which occurs in conjunction with other symptoms such as fever, nausea, vomiting, changes in bowel movements, tenderness, decreased blood pressure, fainting or blood in the feces, you need to seek immediate medical attention.
Anticholinergics should be used with caution in the elderly, in patients with autonomic nervous system disorders, cardiac tachyarrhythmias, arterial hypertension, congestive heart failure, hyperthyroidism and in those with liver and kidney disease. risk of complications related to an excessive anticholinergic effect, care should be taken in patients subject to acute angle glaucoma as well as in patients susceptible to intestinal and urinary stasis and in those prone to tachyarrhythmias.
Due to the possibility that anticholinergics may reduce sweating, Buscopan should be administered with caution to patients with pyrexia.
Interactions Which drugs or foods may change the effect of Buscopan
Tell your doctor or pharmacist if you have recently taken any other medicines, even those without a prescription.
The anticholinergic effect of drugs such as tri- and tetra-cyclic antidepressants, phenothiazines, butyrophenones, antihistamines, antipsychotics, quinidine, amantadine, diisopyramide and other anticholinergics (eg tiotropium, ipratropium and compounds similar to atropine) can be accentuated by Buscopan .
Concomitant treatment with dopamine antagonists, such as metoclopramide, may result in a reduced effect of both drugs on the gastrointestinal tract.
Ss-adrenergic drug-induced tachycardia may be accentuated by Buscopan.
Do not drink alcohol during therapy.
Since antacids can reduce intestinal absorption of anticholinergics, these drugs should not be administered simultaneously.
Warnings It is important to know that:
Anticholinergics can prolong gastric emptying time and cause stasis of the antrum.
Treatment with high doses should not be abruptly stopped. Minor side effects can be controlled by appropriately reducing the dose; the appearance of important secondary manifestations requires the interruption of therapy.
What to do during pregnancy and breastfeeding
Ask your doctor or pharmacist for advice before taking any medicine.
There are limited data from the use of hyoscine N-butylbromide in pregnant women.
Animal studies do not indicate direct or indirect harmful effects with respect to reproductive toxicity.
There is insufficient information on the excretion of Buscopan and its metabolites in human milk. As a precautionary measure, it is preferable to avoid the use of Buscopan during pregnancy and lactation.
Fertility Information
No studies have been conducted to investigate the effects on human fertility.
Effects on ability to drive and use machines
No studies on the ability to drive and use machines have been performed.
Anticholinergics can induce disturbances of visual accommodation and drowsiness, this must be taken into account by those who drive vehicles or machinery or carry out work for which integrity of the degree of vigilance is required.
Important information about some of the ingredients
One 10 mg coated tablet contains 41.2 mg of sucrose corresponding to 247.2 mg per maximum recommended daily dose. So if you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this medicine.
Dose, Method and Time of Administration How to use Buscopan: Posology
How many
The following dosages are recommended for adults and children over the age of 14.
Coated tablets
1-2 coated tablets 3 times a day.
Suppositories
1 suppository 3 times a day.
Individual doses can be increased according to the doctor's judgment.
In pediatrics in children between the ages of 6 and 14, the doctor's prescription must be followed exactly.
In the case of the elderly, in patients with disorders of the autonomic nervous system, in cardiac tachyarrhythmias, in arterial hypertension, in congestive heart failure, in hyperthyroidism and in those with liver and kidney diseases, medical attention should be sought. Caution should also be used. in patients prone to acute angle glaucoma as well as in patients susceptible to intestinal and urinary stasis (see "Precautions for use").
Warning: do not exceed the indicated doses without medical advice.
When and for how long
Warning: use only for short periods of treatment.
Consult your doctor if the spasms recur repeatedly or if you have noticed any recent changes in their characteristics.
Buscopan should not be taken daily on a regular basis or for prolonged periods without researching the cause of abdominal pain.
Like
The tablets should be taken whole with an adequate amount of water.
Overdose What to do if you have taken too much Buscopan
Symptoms
Anticholinergic effects (such as urinary retention, dry mouth, red skin, tachycardia, inhibition of gastrointestinal motility and transient visual disturbances) may occur in the event of overdose.
Therapy
If necessary, administer parasympathomimetic drugs. In case of glaucoma it is necessary to urgently refer to an ophthalmology specialist. Cardiovascular complications must be treated according to the usual therapeutic principles.
In case of respiratory paralysis: consider the opportunity to resort to intubation, artificial respiration. In case of orthostatic hypotension, it is sufficient for the patient to lie down. Catheterization may be necessary for urinary retention.
In addition, if necessary, appropriate supportive care should be undertaken.
In case of accidental ingestion / intake of an overdose of Buscopan, notify your doctor immediately or go to the nearest hospital.
IF IN ANY DOUBT ABOUT USING BUSCOPAN, CONTACT YOUR DOCTOR OR PHARMACIST.
Side Effects What are the side effects of Buscopan
Like all medicines, Buscopan can cause side effects, although not everybody gets them.
Many of the listed side effects can be attributed to the anticholinergic properties of Buscopan. The anticholinergic side effects of Buscopan are generally mild and self-limiting.
Immune system disorders:
Frequency uncommon: skin reactions, urticaria, pruritus.
Frequency not known *: anaphylactic shock, anaphylactic reactions, dyspnoea, skin rash, erythema and other manifestations of hypersensitivity.
* These adverse reactions were observed in post-marketing experience. At 95% probability, the frequency category is not greater than uncommon (3/1368), but may be lower. An accurate estimate of the frequency is not possible since these adverse reactions did not occur in 1368 patients in clinical trials.
Cardiac disorders:
Uncommon frequency: tachycardia.
Gastrointestinal disorders:
Uncommon frequency: dry mouth. Constipation has also been observed.
Skin and subcutaneous tissue disorders: Frequency uncommon: changes in sweating.
Renal and urinary disorders: Frequency rare: urinary retention.
The following side effects have also been observed:
Eye disorders: mydriasis, accommodation disturbances, increased ocular tone. Nervous system disorders: drowsiness.
High doses can lead to signs of central stimulation and more serious signs of interference with the nervous system, state of consciousness and cardiorespiratory function.
Compliance with the instructions contained in the package leaflet reduces the risk of undesirable effects.
Reporting of side effects
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. Side effects can also be reported directly via the national reporting system at https://www.aifa.gov.it/content/segnalazioni-reazioni-avverse. By reporting side effects you can help provide more information on safety of this medicine.
Expiry and Retention
Expiry: see the expiry date printed on the package.
Warning: do not use the medicine after the expiry date shown on the package.
The expiry date refers to the product in intact packaging, correctly stored. Suppositories: do not store above 30 ° C.
Keep this medicine out of the reach and sight of children.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. This will help protect the environment.
It is important to always have the information about the medicine available, so keep both the box and the package leaflet.
Source Package Leaflet: AIFA (Italian Medicines Agency). Content published in January 2016. The information present may not be up-to-date.
To have access to the most up-to-date version, it is advisable to access the AIFA (Italian Medicines Agency) website. Disclaimer and useful information.
01.0 NAME OF THE MEDICINAL PRODUCT
BUSCOPAN COATED TABLETS / SUPPOSITORIES
02.0 QUALITATIVE AND QUANTITATIVE COMPOSITION
Coated tablets
One coated tablet contains: 10 mg hyoscine butylbromide.
Excipients: sucrose.
Suppositories
One suppository contains: hyoscine N-butylbromide 10 mg.
For the full list of excipients see section 6.1.
03.0 PHARMACEUTICAL FORM
Coated tablet.
Suppository.
04.0 CLINICAL INFORMATION
04.1 Therapeutic indications
Symptomatic treatment of painful spastic manifestations of the gastrointestinal and genitourinary tract.
04.2 Posology and method of administration
Dosage
The following dosages are recommended for adults and children over the age of 14.
Coated tablets
1-2 coated tablets 3 times a day.
Suppositories
1 suppository 3 times a day.
Single doses can be increased according to the physician's judgment.
In pediatrics in children between the ages of 6 and 14, the doctor's prescription must be followed exactly.
Method of administration
The tablets should be taken whole with an adequate amount of water.
Buscopan should not be taken daily on a regular basis or for prolonged periods without researching the cause of abdominal pain.
04.3 Contraindications
• Hypersensitivity to the active substance or to any of the excipients.
• Acute angle glaucoma.
• Prostatic hypertrophy or other causes of urinary retention.
• Pyloric stenosis and other conditions stenosing the gastrointestinal canal.
• Paralytic ileus, ulcerative colitis, megacolon.
• Reflux esophagitis.
• Intestinal atony of the elderly and debilitated subjects.
• Myasthenia gravis.
• Children under the age of 6.
In case of rare hereditary conditions of incompatibility with one of the excipients (see section 4.4 "Special warnings and precautions for use") the use of the medicinal product is contraindicated.
04.4 Special warnings and appropriate precautions for use
If you have severe abdominal pain of which the cause is not known, which persists or worsens, or which occurs in conjunction with other symptoms such as fever, nausea, vomiting, changes in bowel movement, abdominal tenderness, decreased blood pressure, fainting or blood in the feces, you need to seek immediate medical attention.
Anticholinergics should be used with caution in the elderly, in patients with disorders of the autonomic nervous system, in cardiac tachyarrhythmias, in arterial hypertension, in congestive heart failure, in hyperthyroidism and in those with liver and kidney disease.
Due to the potential risk of complications related to an excessive anticholinergic effect, caution should be exercised in patients prone to acute angle glaucoma as well as in patients susceptible to intestinal and urinary stasis and in those prone to tachyarrhythmias..
Anticholinergics can prolong gastric emptying time and cause stasis of the antrum.
Due to the possibility that anticholinergics may reduce sweating, Buscopan should be administered with caution in patients with pyrexia.
Treatment with high doses should not be abruptly stopped. Minor side effects can be controlled by appropriately reducing the dose; the appearance of important secondary manifestations requires the interruption of therapy.
One 10 mg coated tablet contains 41.2 mg of sucrose corresponding to 247.2 mg per maximum recommended daily dose. Therefore patients with rare hereditary problems of fructose intolerance should not take this medicine.
04.5 Interactions with other medicinal products and other forms of interaction
The anticholinergic effect of drugs such as tri- and tetracyclic antidepressants, phenothiazines, butyrophenones, antihistamines, antipsychotics, quinidine, amantadine, diisopyramide and other anticholinergics (eg tiotropium, ipratropium and compounds similar to atropine) can be accentuated by Buscopan.
Concomitant treatment with dopamine antagonists, such as metoclopramide, may result in a reduced effect of both drugs on the gastrointestinal tract.
Β-adrenergic drug-induced tachycardia may be accentuated by Buscopan.
Do not drink alcohol during therapy.
Since antacids can reduce intestinal absorption of anticholinergics, these drugs should not be administered simultaneously.
04.6 Pregnancy and lactation
There are limited data from the use of hyoscine N-butylbromide in pregnant women.
Animal studies do not indicate direct or indirect harmful effects with respect to reproductive toxicity (see section 5.3).
There is insufficient information on the excretion of Buscopan and its metabolites in human milk.
As a precautionary measure, it is preferable to avoid the use of Buscopan during pregnancy and breastfeeding.
Studies on the effects on human fertility have not been conducted (see section 5.3).
04.7 Effects on ability to drive and use machines
No studies on the ability to drive and use machines have been performed.
Anticholinergics can induce disturbances of visual accommodation and drowsiness, this must be taken into account by those who drive vehicles or machinery or carry out work for which integrity of the degree of vigilance is required.
04.8 Undesirable effects
Many of the listed side effects can be attributed to the anticholinergic properties of Buscopan. The anticholinergic side effects of Buscopan are generally mild and self-limiting.
Disorders of the immune system:
Frequency uncommon: skin reactions, urticaria, pruritus.
Frequency not known *: anaphylactic shock, anaphylactic reactions, dyspnoea, skin rash, erythema and other manifestations of hypersensitivity.
*These adverse reactions were observed in post-marketing experience. At 95% probability, the frequency category is not greater than uncommon (3/1368), but may be lower. An accurate estimate of the frequency is not possible from since these adverse reactions did not occur in 1368 patients in clinical trials.
Cardiac pathologies:
Uncommon frequency: tachycardia.
Gastrointestinal disorders:
Uncommon frequency: dry mouth.
Constipation has also been observed.
Skin and subcutaneous tissue disorders:
Uncommon frequency: changes in sweating.
Renal and urinary disorders:
Frequency rare: urinary retention.
The following side effects have also been observed:
Eye disorders: mydriasis, accommodation disturbances, increased ocular tone.
Nervous system disorders: sleepiness.
High doses can cause signs of central stimulation and more serious signs of interference with the nervous system, state of consciousness and cardiorespiratory function.
Reporting of suspected adverse reactions
Reporting of suspected adverse reactions occurring after authorization of the medicinal product is important as it allows continuous monitoring of the benefit / risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system. "address https://www.aifa.gov.it/content/segnalazioni-reazioni-avverse.
04.9 Overdose
Symptoms
Anticholinergic effects (such as urinary retention, dry mouth, red skin, tachycardia, inhibition of gastrointestinal motility and transient visual disturbances) may occur in the event of overdose.
Therapy
If necessary, administer parasympathomimetic drugs. In case of glaucoma it is necessary to urgently refer to an ophthalmology specialist. Cardiovascular complications must be treated according to the usual therapeutic principles.
In case of respiratory paralysis: consider the opportunity to resort to intubation, artificial respiration. In case of orthostatic hypotension, it is sufficient for the patient to lie down. Catheterization may be necessary for urinary retention.
In addition, if necessary, appropriate supportive care should be undertaken.
05.0 PHARMACOLOGICAL PROPERTIES
05.1 Pharmacodynamic properties
Pharmacotherapeutic group: semi-synthetic Belladonna alkaloids, quaternary ammonium compounds - hyoscine N-butylbromide (butylscopolamine).
ATC code: A03BB01
Buscopan exerts a spasmolytic action on the smooth muscles of the gastrointestinal, biliary and genitourinary tract.
Being a quaternary ammonium derivative, at the recommended therapeutic doses, hyoscine N-butylbromide does not enter the central nervous system and therefore the undesirable effects on the CNS due to anticholinergics do not occur. The peripheral anticholinergic effect is due to both blocking of the ganglia located in the visceral wall which has antimuscarinic activity.
05.2 Pharmacokinetic properties
Absorption
As a quaternary ammonium, hyoscine N-butylbromide is very polar and is therefore only partially absorbed when administered orally (8%) or rectally (3%).
After oral administration of a single dose of hyoscine N-butylbromide in the range of 20 to 400 mg, peak plasma concentrations between 0.11 ng / mL and 2.04 ng / were recorded after approximately 2 hours. mL.
At the same dose range, the mean observed AUC0-tz values ranged from 0.37 to 10.7 ng.h / mL.
The median value of the absolute bioavailability of the different pharmaceutical forms, ie coated tablets and suppositories, is less than 1%.
Distribution
Given its high affinity with muscarinic and nicotinic receptors, hyoscine N-butylbromide is mainly distributed both in the muscle tissue cells of the abdominal and pelvic area and in the intramural ganglia of the abdominal organs. The plasma protein (albumin) binding of hyoscine N-butylbromide is approximately 4.4%. Animal studies have shown that hyoscine N-butylbromide does not cross the blood-brain barrier, but no clinical data are available. about. Interaction was observed between hyoscine N-butylbromide and choline transport in human placental epithelial cells in vitro.
Metabolism and elimination
After oral administration of single doses in the range of 100 to 400 mg, the half-life of the terminal elimination phase is between 6.2 and 10.6 hours. The major metabolic pathway is hydrolytic cleavage of the ester bond. L " Orally administered hyoscine butylbromide is excreted in the faeces and urine. Studies in humans have shown that 2-5% of the radiolabelled dose is eliminated by the kidney after oral administration and 0.7-1.6% after rectal administration. Approximately 90% of the recovered radioactivity is found in faeces. after oral administration. Renal excretion of hyoscine N-butylbromide is less than 0.1% of the administered dose. The mean apparent oral clearance after oral administration of doses of 100 to 400 mg is between 881 and 1420 L / min, while the corresponding volume of distribution for the same dose range ranges from 6.13 to 11.3 x 105L, probably due to low systemic availability.
Renally excreted metabolites bind weakly to muscarinic receptors and are therefore not thought to contribute to the hyoscine N-butylbromide effect.
05.3 Preclinical safety data
Toxicity
In acute cases, hyoscine N-butylbromide has a low toxicity index: oral LD50 values are 1000-3000 mg / kg in mice, 1040-3300 mg / kg in rat and 600 mg / kg in dogs. Symptoms of toxicity are ataxia and decreased muscle tone and also tremor and convulsions in mice, mydriasis, dry mucous membranes and tachycardia in dogs. Cases of death from respiratory paralysis occurred within 24 hours. LD50 values Intravenous hyoscine N-butylbromide were 10-23 mg / kg in mice and 18 mg / kg in rats. In the 4-week oral repeat-dose toxicity studies, rats tolerated a NOAEL ( no observed adverse effect level) of 500 mg / kg. At doses of 2000 mg / kg, due to the activity of hyoscine N-butylbromide on the parasympathetic ganglia of the visceral wall, the drug caused gastrointestinal paralysis up to constipation. 11 out of 50 rats died.Hematological and clinical laboratory data have no mo dose-related variations. In studies at 26 weeks, rats tolerated doses of 200 mg / kg, while at 250 and 1000 mg / kg gastrointestinal function was depressed and death followed. In studies conducted in dogs of 39 weeks duration, a NOAEL following oral administration (capsules) of 30 mg / kg was recorded. The main clinical manifestations were attributable to the acute effect of high-dose hyoscine N-butylbromide (200 mg / kg). No histopathological changes were observed.
Repeated doses of 1 mg / kg, intravenously, were well tolerated by rats for 4 weeks. At the 3 mg / kg dose, seizures occurred soon after i.v. Rats treated with doses of 9 mg / kg died of respiratory paralysis. Dogs treated for i.v. with doses of 2 x 1, 2 x 3 and 2 x 9 mg / kg for 5 weeks showed mydriasis as a function of the dose administered and for the dose 2 x 9 mg / kg also ataxia, salivation, decrease in body weight and quantity of food taken. Local solutions were well tolerated.
After repeated intramuscular administration, the 10 mg / kg dose was well tolerated systemically, but more muscle lesions at the injection site were found locally in treated rats than in controls. At 60 and 120 mg / kg, mortality was high and the damage observed locally increased with the dose administered.
Hyoscine N-butylbromide proved non-embryotoxic or teratogenic at oral doses up to 200 mg / kg, taken with the diet (rat) or with forced administration by probe of doses up to 200 mg / kg or up to 50 mg / kg subcutaneously (rabbit).
Fertility was not affected by doses up to 200 mg / kg orally.
Like other cationic molecules, hyoscine N-butylbromide interacts with the choline transport system of human placental epithelial cells in vitro. The passage of hyoscine N-butylbromide into the fetal compartment has not been demonstrated.
Hyoscine N-butylbromide in suppositories was locally well tolerated.
In local tolerance studies, dogs and monkeys were treated with repeated intramuscular injections of 15 mg / kg for 28 days: small focal necrosis at the injection site were found only in dogs.
Buscopan was well tolerated when injected into the arteries and veins of the rabbit's ear.
In vitro, a 2% solution for injection of Buscopan showed no haemolytic power when mixed with 0.1 ml of human blood.
Buscopan did not show mutagenic or clastogenic potential in the Ames test, the in-vitro mammalian gene mutation test V79 (HPRT test) and the in-vitro chromosomal aberration test on peripheral human lymphocytes.
In vivo, hyoscine N-butylbromide gave a negative result in the test for the determination of micronuclei in the bone marrow of rats.
There are no in vivo carcinogenicity studies, however hyoscine N-butylbromide showed no carcinogenic potential in two 26-week studies conducted at up to 1000 mg / kg orally in rats.
06.0 PHARMACEUTICAL INFORMATION
06.1 Excipients
Coated tablets:
Core: calcium hydrogen phosphate, corn starch, soluble starch, anhydrous colloidal silica, tartaric acid, stearic acid.
Coating: povidone, sucrose, talc, gum arabic, titanium dioxide (E171), macrogol 6000, carnauba wax, white wax.
Suppositories:
Solid semisynthetic glycerides.
06.2 Incompatibility
Incompatibilities with other medicines are unknown.
06.3 Period of validity
5 years
06.4 Special precautions for storage
Coated tablets: This medicine does not require any special storage conditions.
Suppositories: do not store above 30 ° C.
06.5 Nature of the immediate packaging and contents of the package
Coated tablets: carton containing 2 or 3 aluminum / opaque PVC blisters of 10 coated tablets.
Suppositories: box containing 1 aluminum / PE strip of 6 suppositories.
Not all pack sizes may be marketed.
06.6 Instructions for use and handling
No special instructions.
Unused medicine and waste derived from this medicine must be disposed of in accordance with local regulations.
07.0 MARKETING AUTHORIZATION HOLDER
Boehringer Ingelheim Italia S.p.A.
Via Lorenzini, 8
20139 Milan.
08.0 MARKETING AUTHORIZATION NUMBER
BUSCOPAN 10 mg coated tablets: 20 coated tablets A.I.C. n. 006979013
BUSCOPAN 10 mg coated tablets: 30 coated tablets A.I.C. n. 006979025
BUSCOPAN 10 mg suppositories: 6 A.I.C. n. 006979049
09.0 DATE OF FIRST AUTHORIZATION OR RENEWAL OF THE AUTHORIZATION
20 coated tablets 06.09.1952
30 coated tablets 11.06.1973
6 suppositories 06.09.1952
Renewal
20 coated tablets 1.06.2010
30 coated tablets 1.06.2010
6 suppositories 1.06.2010
10.0 DATE OF REVISION OF THE TEXT
AIFA determination of 24 July 2013