Active ingredients: Repaglinide
NovoNorm 0.5 mg tablets
NovoNorm 1 mg tablets
NovoNorm 2 mg tablets
Indications Why is Novonorm used? What is it for?
NovoNorm is an oral antidiabetic agent containing repaglinide which helps the pancreas to produce more insulin and thereby reduce the level of sugar (glucose) in the blood.
Type 2 diabetes is a disease in which the pancreas does not produce enough insulin to control the level of sugar in the blood or in which the body does not respond normally to the insulin it produces.
NovoNorm is used to control type 2 diabetes in adults as an adjunct to diet and exercise; treatment is usually started when diet, exercise and weight reduction alone are not enough to control ( or reduce) blood glucose levels. NovoNorm can also be given in combination with metformin, another diabetes medicine.
NovoNorm has been shown to reduce blood sugar levels and this helps prevent the complications of diabetes.
Contraindications When Novonorm should not be used
Do not take NovoNorm
- if you are allergic to repaglinide or any of the other ingredients contained in this medicine
- if you have type 1 diabetes.
- if the acid level in the body is high (diabetic ketoacidosis).
- if you have severe liver disease.
- if you take gemfibrozil (medicine used to lower the level of fats in the blood).
Precautions for use What you need to know before taking Novonorm
Talk to your doctor before taking NovoNorm:
- If you have liver problems. NovoNorm is not recommended in patients who have moderate liver disease. NovoNorm must not be taken if you have severe liver disease (see Do not take NovoNorm).
- If you have kidney problems. NovoNorm should be taken with caution.
- If you are about to have major surgery or if you have recently had a serious illness or infection. In these cases it is possible that the diabetes is no longer under control.
- NovoNorm is not recommended if you are under 18 or over 75. NovoNorm has not been studied in these age groups.
Talk to your doctor if any of the above apply to you, NovoNorm may not be suitable for you. Your doctor will inform you.
Children and adolescents
Do not take this medicine if you are under 18 years of age.
If you have hypo (low blood sugar)
If the blood sugar level drops too low, it can go into a hypo (short for hypoglycemia). This can happen:
- If you have taken too much NovoNorm
- If you exercise more than usual
- If you have taken other medicines or suffer from liver or kidney problems (see other sections of the section What you need to know before you take NovoNorm).
The warning signs of a hypo may come on suddenly and can include: cold sweat; cold, pale skin; headache; rapid heartbeat; feeling unwell; strong sense of hunger; temporary visual disturbances; drowsiness; unusual tiredness and weakness; nervousness or tremor; anxiety; confusional state; difficulty concentrating.
If your blood sugar is low or if you feel a hypo is coming: Eat sugar cubes or a high sugar snack or drink, then rest.
When the symptoms of hypoglycaemia disappear or when the blood glucose has stabilized continue treatment with NovoNorm.
Explain to people that you are diabetic and that if you faint (lose consciousness) from a hypo, they should turn you on your side and seek immediate medical attention. They must not give you food or drink. They could suffocate you. .
- If severe hypoglycemia is not treated, it can cause brain damage (temporary or permanent) and even death.
- If you have a hypo that makes you pass out, or a lot of hypos, talk to your doctor. Your NovoNorm dose, diet or type of exercise may need to be changed.
If your blood sugar gets too high
The level of sugar in the blood may become too high (hyperglycaemia). This can happen:
- If you have taken too little NovoNorm
- If you have an infection or fever
- If you have eaten more than usual
- If you have exercised less than usual
The warning signs of too high blood sugar appear gradually. They include: increased diuresis; thirst; dryness of the skin and mouth. Consult your doctor. The amount of NovoNorm you take, your diet or exercise may need to be changed.
Interactions Which drugs or foods can change the effect of Novonorm
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines.
You can take NovoNorm in combination with metformin, another diabetes medicine, if your doctor prescribes it.
If you are taking gemfibrozil (used to lower blood fat levels) you should not take NovoNorm.
The body's response to NovoNorm may vary if you take other medicines, in particular :.
- Monoamine oxidase inhibitors (MAOIs, used to treat depression)
- Beta blockers (used to treat high blood pressure and heart disease)
- ACE inhibitors (used to treat heart disease)
- Salicylates (e.g. aspirin)
- Octreotide (used to treat cancer)
- Non-steroidal anti-inflammatory drugs (NSAIDs, a type of pain reliever)
- Steroids (anabolic steroids and corticosteroids - used for anemia or to treat inflammation)
- Oral contraceptives (used for birth control)
- Thiazides (diuretics or "water pills")
- Danazol (used to treat breast cysts and endometriosis)
- Thyroid hormones (used in case of low thyroid hormone levels)
- Sympathomimetics (used to treat asthma)
- Clarithromycin, trimethoprim, rifampicin (antibiotics)
- Itraconazole, ketoconazole (medicine against fungal infections)
- Gemfibrozil (used to lower blood fat levels)
- Ciclosporin (used to suppress the immune system)
- Deferasirox (used to reduce chronically high iron level)
- Phenytoin, carbamazepine, phenobarbital (used to treat epilepsy)
- St. John's wort (herbal medicine).
NovoNorm with alcohol
NovoNorm's ability to lower blood sugar may vary if you drink alcohol. Look out for signs of a "hypoglycemia."
Warnings It is important to know that:
Pregnancy and breastfeeding
If you are pregnant or breastfeeding, suspect pregnancy or plan to become pregnant, please tell your doctor before taking this medicine.
You must not take NovoNorm if you are pregnant or planning to become pregnant.
You should not take NovoNorm if you are breastfeeding.
Driving and using machines
The ability to drive or use machines may be affected by low or high blood sugar. Keep in mind that it could endanger yourself and others. Ask your doctor if you can drive a vehicle if:
- undergoes frequent hypoglycemic episodes
- he does not feel the warning signs of hypoglycemia or feels few of them
Dose, Method and Time of Administration How to use Novonorm: Posology
Always use this medicine exactly as your doctor has told you.
If in doubt, consult your doctor. Your doctor will work out the dose for you.
- The starting dose is usually 0.5 mg before each main meal. Swallow the tablets with a glass of water just before each main meal or within 30 minutes before the meal.
- The dose can be changed by your doctor up to a maximum of 4 mg taken immediately before each main meal or in the 30 minutes before the meal. The maximum recommended daily dose is 16 mg. Do not take doses of NovoNorm higher than those prescribed by your doctor.
Overdose What to do if you have taken too much Novonorm
If you take more NovoNorm than you should
If you take too many tablets, your blood sugar can become too low, leading to a hypo. See If you have a hypo, what a hypo is for and how to treat it.
If you forget to take NovoNorm
If you miss a dose, take the next one as usual, but don't double it.
If you stop taking NovoNorm
Note that if you stop taking NovoNorm the desired effect is not achieved. Your diabetes may get worse. Contact your doctor first if you need to change your treatment.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
Side Effects What are the side effects of Novonorm
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Hypoglycemia
The most common side effect is hypoglycaemia which may affect up to 1 in 10 patients (see If you have a hypo in section 2). Hypoglycaemic reactions are usually mild / moderate but may occasionally progress to unconsciousness or a hypoglycemic coma. In this case, contact medical assistance immediately.
Allergy
Allergy is very rare (may affect up to 1 in 10,000 people). Symptoms such as swelling, difficulty breathing, fast heartbeat, dizziness and sweating may be signs of an anaphylactic reaction. Contact a doctor immediately.
Other side effects
Common (may affect up to 1 in 10 patients)
- Stomach ache
- Diarrhea.
Rare (may affect up to 1 in 1,000 patients)
- Acute coronary syndrome (but this effect may not be drug related).
Very rare (may affect up to 1 in 10,000 patients)
- He retched
- Constipation
- Visual disturbances
- Severe liver problems, abnormal liver function, increase in liver enzymes in the blood.
Frequency not known
- Hypersensitivity (such as rash, itching, redness of the skin, swelling of the skin)
- Malaise (nausea)
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet.
Expiry and Retention
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the carton and blister after EXP. The expiry date refers to the last day of the month indicated.
Store in the original package to protect from moisture.
Do not throw any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. This will help protect the environment.
What NovoNorm contains
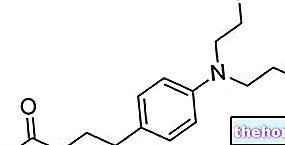
- The active ingredient is repaglinide.
- The other ingredients are microcrystalline cellulose (E460), anhydrous monohydrogenated calcium phosphate, maize starch, amberlite (polacrylin potassium), povidone (polyvidone), glycerol 85%, magnesium stearate, meglumine and poloxamer, yellow iron oxide (E172) only in 1 mg tablets and red iron oxide (E172) in 2 mg tablets only.
What NovoNorm looks like and contents of the pack
NovoNorm tablets are round, convex and embossed with the Novo Nordisk logo (Bee Bee). Concentrations are 0.5 mg, 1 mg and 2 mg. The 0.5 mg tablets are white, the 1 mg tablets are yellow and the 2 mg tablets are peach. 4 blister packs are available. Each pack contains 30, 90, 120, or 270 tablets.
Not all pack sizes may be marketed.
Source Package Leaflet: AIFA (Italian Medicines Agency). Content published in January 2016. The information present may not be up-to-date.
To have access to the most up-to-date version, it is advisable to access the AIFA (Italian Medicines Agency) website. Disclaimer and useful information.
01.0 NAME OF THE MEDICINAL PRODUCT
NOVONORM 0.5 MG TABLETS
02.0 QUALITATIVE AND QUANTITATIVE COMPOSITION
Each tablet contains 0.5 mg of repaglinide.
For the full list of excipients, see section 6.1.
03.0 PHARMACEUTICAL FORM
Tablets
Repaglinide tablets are white, round, convex and stamped with the Novo Nordisk logo (Bee Bee).
04.0 CLINICAL INFORMATION
04.1 Therapeutic indications
Repaglinide is indicated for adults with type 2 diabetes mellitus whose hyperglycaemia cannot be satisfactorily controlled by diet and exercise. Repaglinide is also indicated in combination with metformin in adults with type 2 diabetes mellitus who are not satisfactorily controlled on metformin alone.
Treatment should be started in addition to diet and exercise to reduce meal-related blood glucose levels.
04.2 Posology and method of administration
Dosage
Repaglinide is given before meals and dosed individually to optimize glycemic control. The treating physician must periodically check blood glucose to establish the minimum effective dose for the individual patient, in addition to the patient's normal home self-monitoring of blood glucose and / or glucosuria. Glycosylated hemoglobin levels can also be used to control therapeutic response. It is necessary to carry out periodic blood glucose monitoring to identify cases in which an adequate reduction in blood glucose levels has not been achieved despite the administration of maximum doses of the drug (primary failure) and to identify cases in which there is a loss of the ability to adequately control blood glucose after an initial period in which the drug has been effective (secondary failure).
Short-term administration of repaglinide may be sufficient for transient loss of glycemic control in patients with type 2 diabetes who are normally well compensated by diet alone.
Initial dose
The dosage should be determined by the attending physician based on the patient's needs.
The recommended starting dose is 0.5 mg. Approximately one to two weeks should elapse between dose adjustments (based on glycemic response).
If patients are transferred from another oral hypoglycemic agent, the recommended starting dose is 1 mg.
Maintenance
The maximum recommended single dose is 4 mg taken with main meals.
The maximum total daily dose should not exceed 16 mg.
Special patient groups
Senior citizens
No clinical studies have been performed in patients over 75 years old.
Kidney failure
Repaglinide is mainly excreted via the biliary route and is therefore not susceptible to kidney disease.
8% of a dose of repaglinide is excreted via the kidneys and plasma clearance of the product is reduced in patients with renal insufficiency. Since insulin sensitivity is higher in diabetics with renal insufficiency, caution should be exercised in adjusting the dose in these patients.
Hepatic insufficiency
No clinical studies have been performed in patients with hepatic insufficiency.
Debilitated or malnourished patients
In debilitated or malnourished patients, the initial and maintenance dose should be conservative and careful dose adjustment is required in order to avoid hypoglycaemic reactions.
Patients treated with other oral hypoglycemic agents
Patients treated with other oral hypoglycaemics can switch directly to repaglinide treatment, although there is no "exact dose relationship between repaglinide and other oral hypoglycaemics. The maximum recommended starting dose for patients switching to repaglinide is 1 mg to be taken immediately before main meals.
Repaglinide can be given in combination with metformin when blood glucose is not sufficiently controlled with metformin alone. In this case, the metformin dosage should be left unchanged while simultaneously administering repaglinide. The starting dose of repaglinide is 0.5 mg before main meals; dosage adjustment should be established on the basis of glycemic response as for monotherapy.
Pediatric population
The safety and efficacy of repaglinide in children below 18 years have not been established. No data are available.
Method of administration
Repaglinide should be taken immediately before main meals (i.e. pre-meal administration).
The doses are usually taken about 15 minutes before a meal but the time can vary from immediately before to 30 minutes before the meal (before 2, 3 or 4 meals a day). Patients who skip a meal (or have an extra meal) should be instructed to skip (or add) a dose in relation to that meal.
In case of concomitant use with other active substances refer to sections 4.4 and 4.5 to determine the dose.
04.3 Contraindications
• Known hypersensitivity to repaglinide or to any of the excipients listed in section 6.1.
• Type 1 diabetes mellitus, C negative peptide.
• Diabetic ketoacidosis, with or without coma.
• Severe liver dysfunction.
• Concomitant use of gemfibrozil (see section 4.5).
04.4 Special warnings and appropriate precautions for use
General
Repaglinide should only be prescribed if insufficient glycemic control and symptoms of diabetes persist despite adequate attempts at diet, physical activity and weight reduction.
Hypoglycemia
Repaglinide, like other insulin secretagogues, can cause hypoglycaemia.
Combination with insulin secretagogues
Over time, the ability of an oral hypoglycemic agent to lower blood glucose decreases in many patients. This event may be due to worsening of the diabetes or a reduced ability to respond to the medicine. This situation, known as secondary failure, must be distinguished from primary failure in which the drug is ineffective from the outset. Before classifying a patient as a secondary failure subject, the dose should be adjusted and adherence to diet and exercise assessed. .
Repaglinide acts through a specific binding site with a short action on beta cells. No clinical studies have been performed on the use of repaglinide in cases of secondary failure to insulin secretagogues.
No clinical studies have been performed on the combination with other insulin secretagogues.
Combination with Hagerdon's Neutral Protamine Insulin (NPH) or with thiazolidinediones
Studies have been performed on combination therapy with NPH insulin or with thiazolidinediones. However, the benefit / risk profile in comparison with other combination therapies remains to be defined.
Combination with metformin
Combined treatment with metformin is associated with an increased risk of hypoglycaemia.
When a patient stabilized on any oral hypoglycemic agent experiences stress such as fever, trauma, infection, or surgery, a loss of glycemic control can occur. In such cases, it may be necessary to discontinue repaglinide and transiently treat the patient with insulin.
Acute coronary syndrome
The use of repaglinide may be associated with an increased incidence of acute coronary syndrome (eg myocardial infarction), see sections 4.8 and 5.1.
Concomitant use
Repaglinide should be used with caution or avoided in patients taking medicinal products that affect the metabolism of repaglinide (see section 4.5). If concomitant use is necessary, blood glucose should be closely monitored as well as careful clinical monitoring.
04.5 Interactions with other medicinal products and other forms of interaction
Numerous medicines are known to affect the metabolism of repaglinide, therefore the physician must take into account possible interactions:
Data obtained from studies in vitro indicate that repaglinide is metabolised predominantly by CYP2C8, but also by CYP3A4. Clinical data from healthy volunteers confirm that CYP2C8 is the most important enzyme involved in the metabolism of repaglinide while CYP3A4 plays a minor role, but its relative contribution may be increased if CYP2C8 is inhibited. Consequently, the metabolism, and thereby the clearance of repaglinide, may be altered by substances that affect these cytochrome P-450 enzymes both inhibitory and inductive. Special attention should be paid when both CYP2C8 and 3A4 inhibitors are administered simultaneously with repaglinide.
Based on data obtained from studies in vitro, repaglinide appears to be a substrate for active hepatic uptake (organic anion transporter protein OATP1B1). OATP1B1 inhibitors may increase plasma concentrations of repaglinide, as has been shown for cyclosporine (see below).
The hypoglycemic effect of repaglinide may be increased and / or prolonged by the following substances: gemfibrozil, clarithromycin, itraconazole, ketoconazole, trimethoprim, cyclosporine, other antidiabetic drugs, monoamine oxidase inhibitors, non-selective beta-blockers, inhibitors of " angiotensin converting enzyme (ACE inhibitors), salicylates, NSAIDs, octeotride, alcohol and anabolic steroids.
Concomitant administration of gemfibrozil (600 mg twice daily), an inhibitor of CYP2C8, and repaglinide (a single dose of 0.25 mg), increased the area under the curve (AUC) of the repaglinide and 2.4 times the Cmax in healthy volunteers. The half-life was prolonged from 1.3 to 3.7 hours resulting in a possible increase and prolongation of the blood glucose lowering effect of repaglinide and the plasma concentration of repaglinide at 7 hours increased 28.6-fold from taking gemfibrozil. Concomitant use of gemfibrozil and repaglinide is contraindicated (see section 4.3).
Concomitant administration of trimethoprim (160 mg twice daily), a weak inhibitor of CYP2C8, and repaglinide (a single dose of 0.25 mg) increases the AUC, Cmax and t½ of repaglinide (1.6-fold). , 1.4-fold and 1.2-fold respectively) with no statistically significant effects on blood glucose. This lack of pharmacodynamic effect was observed with a dose of repaglinide below the therapeutic dose. As the safety profile of this combination has not been established with In doses above 0.25 mg for repaglinide and 320 mg for trimethoprim, concomitant use of trimethoprim with repaglinide should be avoided. If concomitant use is necessary, blood glucose should be closely monitored as well as careful clinical monitoring (see section 4.4).
Rifampicin, a potent inducer of CYP3A4 but also CYP2C8, acts as both an inducer and an inhibitor in the metabolism of repaglinide. A seven-day pretreatment with rifampicin (600 mg), followed by concomitant administration of repaglinide (a single dose of 4 mg) on the seventh day decreases the AUC (combined inductive and inhibitory effect) by 50%. When repaglinide was given 24 hours after the last rifampicin dose, an 80% reduction in repaglinide AUC was observed (inductive effect only). Concomitant use of rifampicin and repaglinide may therefore require a dose adjustment of repaglinide to be defined by careful monitoring of blood glucose both at the start of treatment with rifampicin (acute inhibition), and at subsequent doses (combined inhibition and induction) and at discontinuation of treatment (induction only) until approximately two weeks after discontinuation of rifampicin when the inductive effect of rifampicin is no longer present. It cannot be excluded that other inducers eg phenytoin, carbamazepine, phenobarbital and St. John's wort can have a similar effect.
The effect of ketoconazole, a prototype of a potent and competitive inhibitor of CYP3A4, on the pharmacokinetics of repaglinide was studied in normal subjects. Co-administration of ketoconazole 200 mg increases the AUC and Cmax of repaglinide by 1.2-fold. with glycemic profiles altered by less than 8% when administered concomitantly (a single 4 mg dose of repaglinide). Co-administration of 100 mg of itraconazole, a CYP3A4 inhibitor, has also been studied in healthy volunteers and has showed a 1.4-fold increase in AUC. There was no significant effect on glucose levels in healthy volunteers. In a drug interaction study conducted in healthy volunteers, co-administration of 250 mg clarithromycin, a potent mechanism-of-action inhibitor of CYP3A4, slightly increased the AUC of repaglinide by 1.4-fold and the Cmax of. 1.7 times, and increases the mean increase in serum insulin AUC by 1.5 times and the maximum concentration by 1.6 times. The exact mechanism of this interaction is not yet clear.
In a study conducted in healthy volunteers, concomitant administration of repaglinide (a single 0.25 mg dose) and cyclosporine (repeated doses of 100 mg) increased AUC and Cmax by approximately 2.5-fold and 1-fold, respectively. , 8 times. As interaction with repaglinide dosages higher than 0.25 mg has not been established, concomitant use of ciclosporin with repaglinide should be avoided. If this combination is deemed necessary, careful clinical monitoring should be performed and blood glucose (see section 4.4).
In an interaction study conducted in healthy volunteers, co-administration of deferasirox (30 mg / kg / day, 4 days), a weak inhibitor of CYP2C8 and CYP3A4, and repaglinide (single dose, 0.5 mg) resulted in an increase in repaglinide systemic exposure (AUC) up to 2.3 times the control (90% CI [2.03-2.63]), a 1.6 times increase (90% CI [1.42-1.84]) Cmax and a small but significant increase in blood glucose values. Since interaction with doses higher than 0.5 mg of repaglinide has not been established, concomitant use of deferasirox and repaglinide should be avoided. "Combined use, close clinical and blood glucose monitoring should be performed (see section 4.4).
B-blocking drugs can mask the symptoms of hypoglycemia.
Concomitant administration of cimetidine, nifedipine, estrogen or simvastatin with repaglinide, all substrates of CYP3A4, did not significantly alter the pharmacokinetic parameters of repaglinide.
Repaglinide had no clinically relevant effects on the steady-state pharmacokinetic properties of digoxin, theophylline or warfarin when administered to healthy volunteers. Therefore, in case of concomitant administration of repaglinide with these drugs, no dosage adjustments are necessary.
The blood glucose lowering effect of repaglinide can be reduced by the following substances:
oral contraceptives, rifampicin, barbiturates, carbamazepine, thiazides, corticosteroids, danazol, thyroid hormones and sympathomimetics.
When these medicinal products are added to or removed from therapy in a patient treated with repaglinide, the patient should be closely monitored for changes in glycemic control.
A potential interaction should be considered when repaglinide is used with other medicinal products which are also secreted mainly via the bile.
Pediatric population
No interaction studies have been performed in children and adolescents.
04.6 Pregnancy and lactation
Pregnancy
There are no studies regarding the use of repaglinide in pregnant women. Repaglinide should be avoided during pregnancy.
Feeding time
There are no studies regarding the use of repaglinide in breastfeeding women. Repaglinide should not be used during breastfeeding.
Fertility
Animal studies have shown reproductive toxicity (see section 5.3).
04.7 Effects on ability to drive and use machines
NovoNorm has no direct influence on the ability to drive and use machines, but may cause hypoglycaemia.
Advise patients to take the necessary precautions to avoid a hypoglycaemic episode while driving. This is particularly important in those who have little or no awareness of the warning symptoms of hypoglycemia or who have frequent episodes of hypoglycemia.In these circumstances, driving should be discouraged.
04.8 Undesirable effects
Summary of the safety profile
The most frequently reported adverse reactions are changes in blood glucose levels, eg hypoglycaemia. The occurrence of these reactions depends on individual factors, such as eating habits, dosage, exercise and stress.
Table of adverse reactions
Based on experience with repaglinide and other hypoglycaemics, the following adverse reactions have been observed. Frequency is defined as: common (≥1 / 100 to
* see section Description of selected adverse reactions below
Description of selected adverse reactions
Allergic reactions
Generalized hypersensitivity reactions (e.g. anaphylactic reactions), or immunological reactions such as vasculitis.
Disorder of refraction
Changes in blood glucose levels have been observed to cause transient visual disturbances, especially at the start of treatment. These disturbances have only been reported in very rare cases after initiation of repaglinide treatment and have not been reported in clinical trials. never required discontinuation of repaglinide treatment.
Abnormal liver function, increased liver enzymes
Isolated cases of elevated liver enzymes have been reported during treatment with repaglinide. Most cases were mild and transient and only a very few patients were forced to discontinue therapy. In very rare cases, severe hepatic dysfunction has been reported.
Hypersensitivity
Skin hypersensitivity reactions such as erythema, pruritus, skin rash and urticaria may occur. There is, however, no reason to suspect a cross allergy with sulfonylureas due to the diversity in chemical structure.
04.9 Overdose
Repaglinide was administered in weekly dose increases of 4 to 20 mg four times a day for a period of 6 weeks. No relevant data emerged regarding the safety of the drug. Since in this study the onset of hypoglycemia with increased caloric intake was avoided, a relative overdose can cause an excessive reduction in blood sugar with consequent development of hypoglycemic symptoms (dizziness, sweating, tremors, headache, etc.). cases, it is recommended to take appropriate intervention measures to correct the reduction in blood sugar (oral carbohydrates). More severe hypoglycaemia associated with seizures, loss of consciousness or coma should be treated with glucose administered intravenously.
05.0 PHARMACOLOGICAL PROPERTIES
05.1 Pharmacodynamic properties
Pharmacotherapeutic group: other hypoglycemic drugs, excluding insulins. ATC code: A10BX02
Mechanism of action
Repaglinide is a short-acting oral secretagogue. Repaglinide rapidly lowers blood glucose levels by stimulating insulin secretion by the pancreas, an effect that depends on the functioning of pancreatic islet beta cells.
Repaglinide closes the potassium-dependent ATP channels of the β-pancreatic cell membrane through a binding site different from that of other secretagogues. This action depolarizes the beta cells and causes the calcium channels to open. The resulting increase in intracellular calcium flow stimulates the secretion of beta cells.
Pharmacodynamic effects
In patients with type 2 diabetes, insulin secretion in response to meals occurs within 30 minutes of oral administration of repaglinide. This action causes the reduction of blood sugar throughout the period influenced by meals. The increase in insulin levels did not persist beyond the duration of the meal. Plasma levels of repaglinide decreased rapidly, resulting in low concentrations of the drug 4 hours after administration, in the plasma of type 2 diabetics.
Clinical efficacy and safety
In patients with type 2 diabetes, a dose-dependent reduction in blood glucose was seen with doses of repaglinide ranging from 0.5 to 4 mg.
The results of clinical studies have shown that the optimal administration of repaglinide should be done in relation to main meals (pre-meal administration).
Repaglinide is normally taken 15 minutes before a meal, but the time of intake can fluctuate from just before to 30 minutes before a meal.
An epidemiological study has suggested a higher risk of acute coronary syndrome in patients treated with repaglinide than in patients treated with sulfonylurea (see sections 4.4 and 4.8).
05.2 Pharmacokinetic properties
Absorption
Repaglinide is rapidly absorbed from the gastrointestinal tract, causing an equally rapid increase in the plasma concentration of the active substance. The plasma peak occurs within one hour of administration. After reaching the maximum peak, the plasma level rapidly decreases. Repaglinide is characterized by a mean absolute bioavailability of 63% (CV 11%).
No clinically significant differences in repaglinide pharmacokinetics were observed when it was administered 0, 15 or 30 minutes before a meal or under fasted conditions.
In clinical studies, "high interindividual variability in repaglinide plasma concentrations (60%) was found. Intraindividual variability is low or moderate (35%) and, as repaglinide posology must be adjusted based on clinical response," efficacy is not affected by interindividual variability.
Distribution
The pharmacokinetics of repaglinide are characterized by a low volume of distribution, 30 L (compatible with distribution into intercellular fluids) and in humans it is highly bound to plasma proteins (greater than 98%).
Elimination
Repaglinide is rapidly cleared from the blood within 4-6 hours. The plasma elimination half-life is approximately one hour.
Repaglinide is almost completely metabolised and no metabolites with hypoglycemic effects of clinical relevance have been identified.
The metabolites of repaglinide are mainly excreted via the biliary route. A small fraction (less than 8%) of the administered dose appears in the urine, mainly as metabolites. Less than 1% of repaglinide is present in the faeces.
Special patient groups
Administration of repaglinide results in higher plasma concentrations in patients with hepatic insufficiency and elderly type 2 diabetics. The area under the curve (AUC: mean ± SD) after administration of a single dose of 2 mg (4 mg in patients with hepatic insufficiency) was 31.4 ng / ml / hour (± 28.3) in volunteers. healthy, 304.9 ng / ml / hour (± 228.0) in patients with hepatic insufficiency and 117.9 ng / ml / hour (± 83.8) in elderly type 2 diabetics.
After 5 days of treatment with repaglinide (2 mg x 3 times daily) in patients with severe renal insufficiency (creatinine clearance: 20-39 ml / min.), The results showed a significant 2-fold increase in repaglinide concentration ( AUC) and its half-life (t1 / 2) compared to that found in subjects with normal renal function.
Pediatric population
No data are available.
05.3 Preclinical safety data
Non-clinical data revealed no special hazard for humans based on conventional studies on safety pharmacology, repeated dose toxicity, genotoxicity and carcinogenic potential.
In animal studies, repaglinide did not show teratogenic effects. Embryotoxicity, abnormal limb development in fetuses and lactating offspring was observed in female rats exposed to high doses during late pregnancy and during lactation. Repaglinide was detected in the milk of experimental animals.
06.0 PHARMACEUTICAL INFORMATION
06.1 Excipients
Microcrystalline cellulose (E460)
Monohydrogenated calcium phosphate, anhydrous
Cornstarch
Amberlite (polacrilin potassium)
Povidone (polyvidone)
Glycerol 85%
Magnesium stearate
Meglumine
Poloxamer
06.2 Incompatibility
Not relevant.
06.3 Period of validity
5 years.
06.4 Special precautions for storage
Store in the original package to protect from moisture.
06.5 Nature of the immediate packaging and contents of the package
The blister pack (aluminum / aluminum) contains 30, 90, 120 or 270 tablets respectively.
Not all pack sizes may be marketed.
06.6 Instructions for use and handling
No special instructions.
07.0 MARKETING AUTHORIZATION HOLDER
Novo Nordisk A / S
Novo Allé
DK-2880 Bagsværd
Denmark
08.0 MARKETING AUTHORIZATION NUMBER
EU / 1/98/076 / 004-006, EU / 1/98/076/023
034162040
034162053
034162065
09.0 DATE OF FIRST AUTHORIZATION OR RENEWAL OF THE AUTHORIZATION
Date of first authorization: 17 August 1998
Date of last renewal: August 17, 2008
10.0 DATE OF REVISION OF THE TEXT
04/2012