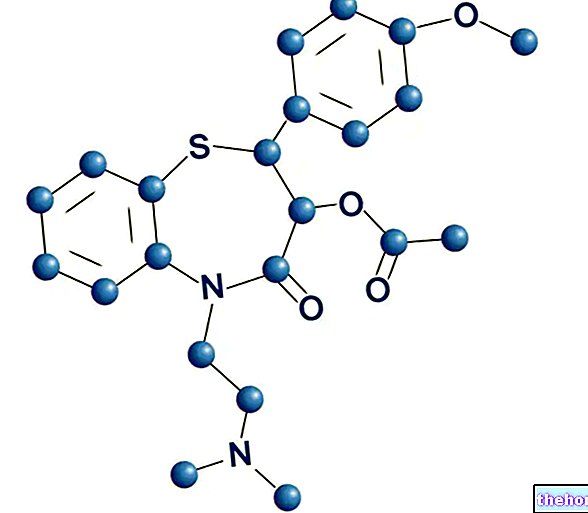
BELARA ® is a drug based on ethinylestradiol + Chlormadinone acetate
THERAPEUTIC GROUP: Systemic hormonal contraceptives - Progestin and estrogen, fixed combination

Indications BELARA ® - Contraceptive Pill
BELARA ® is used as an oral contraceptive.
Mechanism of action BELARA ® - Contraceptive Pill
BELARA ® is an oral contraceptive consisting of low-dose estrogens (ethinylestradiol) and a progestin (chlormadinone) at concentrations useful both to safeguard the contraceptive characteristics and to reduce some unpleasant effects of combined oral contraception.
The anti-contraceptive effect is guaranteed by the synergistic action of the two hormones useful for inhibiting the hypothalamus-pituitary axis, reducing the endogenous secretion of gonadotropins responsible for follicular maturation and subsequent ovulation, and guaranteeing changes in the endometrium and cervical mucus such as to complicate the ascent of the spermatozoa in the female genital tract and the possible nesting of the embryo.
Chlormadinone is also attributed a strong antiandrogenic action useful for avoiding some particularly annoying reactions related to the pill such as acne, hirsutism and alopecia.
Both active ingredients, taken orally, are absorbed in the intestine reaching the maximum plasma concentration in about 2 hours, and persisting in circulation for over 20 hours, allowing the drug to be administered once a day.
Once their action is over, the two hormones contained in BELARA ® first undergo a hepatic metabolism and subsequently an elimination through urine and faeces.
Studies carried out and clinical efficacy
1. ORAL CONTRACEPTION AND MENSTRUAL PAIN
Clin Drug Investig. 2011; 31: 269-77.
Extended cycles with the combined oral contraceptive chlormadinone acetate 2 mg / ethinylestradiol 0.03 mg: pooled analysis of data from three large-scale, non-interventional, observational studies.
Göretzlehner G, Waldmann-Rex S, Schramm GA.
Study that demonstrates how long-term use of oral contraceptives can be useful in reducing painful symptoms associated with the cycle. Important property that demonstrates the therapeutic usefulness of oral contraception.
2. BELARA AND ACNE
Contraception. 2009 Jul; 80: 25-33. Epub 2009 May 9.
Efficacy of an oral contraceptive containing EE 0.03 mg and CMA 2 mg (Belara) in moderate acne resolution: a randomized, double-blind, placebo-controlled Phase III trial.
Plewig G, Cunliffe WJ, Binder N, Höschen K.
The antiandrogenic action of chlormadinone acetate has been shown to be useful in improving moderate acne in patients undergoing contraceptive therapy. This work demonstrates the beneficial effects on the skin of contraceptives such as BELARA.
3. BELARA AND METABOLIC PROFILE
Contraception. 2009 Jan; 79: 15-23. Epub 2008 Nov 17.
The effects of two monophasic oral contraceptives containing 30 mcg of ethinyl estradiol and either 2 mg of chlormadinone acetate or 0.15 mg of desogestrel on lipid, hormone and metabolic parameters.
Winkler UH, Sudik R.
Administration of BELARA was found to be safer than that of other oral contraceptives in maintaining metabolic health. This study has in fact demonstrated how the assumption of the aforementioned contraceptive can preserve the patient's lipid profile by not contributing to the increase in atherosclerotic risk, as is done by other oral contraceptives.
Method of use and dosage
BELARA ® Ethylestradiol 0.03 mg and chlormadinone acetate 2 mg coated tablets:
Before starting to take an oral contraceptive it would be necessary to consult your gynecologist and undergo a careful check-up.
In cases where the administration of BELARA ® is indicated, one should take one tablet per day for 21 days, followed by suspension for 7 days in which withdrawal bleeding similar to the physiological menstruation should occur.
The half-life of the two active ingredients allows to maintain the maximum contraceptive activity through the ingestion of one tablet every day at the same time.
Contraceptive coverage therefore tends to decrease 36 hours after the last tablet taken while the hypothalamus-pituitary axis recovers its physiological activity after about 7 days of interruption.
In the event that the tablet is vomited within 3-4 hours of its intake, it would be necessary to resort to a new administration.
In case of forgetfulness, recent pregnancy or transition from a different contraceptive therapy, it is useful to consult your gynecologist and strictly follow his instructions.
BELARA ® Warnings - Contraceptive Pill
The biological complexity of oral contraception, the numerous side effects and the increased risk of onset of serious diseases such as neoplastic and thromboembolic ones require a careful preliminary check-up, useful to verify the therapeutic appropriateness and the absence of incompatible conditions.
In fact, it is necessary to take into account that in addition to coagulation disorders, cardiovascular diseases, neoplastic diseases, liver and kidney diseases, neurological and psychiatric disorders and metabolic diseases such as diabetes, predisposing conditions such as "obesity or smoking, can significantly affect the" onset of thromboembolic events especially in conjunction with the administration of combined oral contraceptives.
In the aforementioned cases it is therefore necessary a "careful medical supervision and periodic checks.
BELARA ® contains lactose so its intake in patients with lactase enzyme deficiency, glucose / galactose poor absorption or lactose intolerance, could be associated with even serious gastrointestinal disorders.
PREGNANCY AND BREASTFEEDING
The use of BELARA ® in pregnancy is contraindicated despite the fact that the scientific literature has not yet found clinical correlations capable of associating teratogenic effects on the fetus with the administration of combined oral contraceptives.
On the contrary, pharmacokinetic studies have shown that both active ingredients can easily pass the breast filter and concentrate in breast milk, thus extending the contraindication also to the lactation period.
Interactions
Although chlormadinone does not undergo a first pass metabolism, both active principles are metabolized at the end of their life by liver enzymes, and in particular by the cytochromes family.
This type of metabolism exposes the two hormones to particular pharmacokinetic variations, given the high susceptibility of the aforementioned enzymes to exogenous modulators.
In fact, rifampicin, phenytoin, barbiturates, antiretrovirals, antibiotics, herbal remedies such as St. John's wort, represent only some of the active ingredients capable of modulating the activity of cytochromial enzymes, consequently varying the pharmacokinetic properties and the contraceptive efficacy of BELARA. ®
At the same time it must be considered that ethinylestradiol is able to modify the biological activity of other active principles
Therefore, given the complexity of possible interactions, it would be useful to consult your doctor in case of concomitant administration of multiple drugs and to read the package leaflet carefully.
Contraindications BELARA ® - Contraceptive Pill
BELARA ® is contraindicated in case of current or previous venous thrombosis, stroke, hypertension, metabolic diseases such as diabetes mellitus, hypertension and dyslipidemias, impaired liver and kidney function, malignant diseases, neuro-psychiatric disorders, motor disorders, undiagnosed gynecological diseases and in case of hypersensitivity to the active substance or to one of its excipients.
Undesirable Effects - Side Effects
The intake of BELARA ® is accompanied especially in the initial phase of the contraceptive cycle with side effects related to:
- nervous system such as headache, dizziness, migraine and behavioral changes;
- gastrointestinal tract with nausea, vomiting and abdominal pain;
- cardiovascular system with a significant increase in blood pressure;
- genital tract with hemorrhagic spotting, vaginal discharge and pelvic pain;
- breast with increased breast tenderness and pain.
Clinically relevant side effects have been observed in rare cases, while the association between COCs and increased risk of thromboembolic and neoplastic pathologies remains to be clarified.
Note
BELARA ® can only be sold under medical prescription.
The information on BELARA ® - Contraceptive Pill published on this page may be out of date or incomplete. For a correct use of this information, see the Disclaimer and useful information page.